The delicate art of creating ginger milk curd, known in Cantonese as 姜汁撞奶 (gong ji zong nai) or 薑汁燉奶, has long captivated culinary enthusiasts with its seemingly simple three-ingredient composition: milk, ginger, and sugar. This traditional dessert, cherished for its tender, fragile gel texture and distinct spicy-sweet flavor, presents a fascinating paradox. Despite its minimalist recipe, achieving consistent success has proven notoriously difficult for many home cooks, leading to a proliferation of conflicting advice and what can only be described as culinary folklore. Recent scientific investigations, however, are now demystifying this enigmatic dessert, revealing the precise enzymatic reactions at play and offering a blueprint for consistent, fool-proof preparation.
The Enigma of a Three-Ingredient Delicacy
Ginger milk curd stands out in the culinary landscape as a unique example of enzymatic coagulation. Unlike many gels that rely on external hydrocolloids like gelatin or agar, this dessert sets entirely through the action of enzymes naturally present in fresh ginger. The allure lies in its rapid transformation—milk, heated to a specific temperature, is poured over fresh ginger juice and sets into a delicate curd within minutes, often firm enough to support a spoon. This captivating transformation has made it a staple in Southern Chinese cuisine, particularly in Guangdong and Hong Kong, where it is often enjoyed warm as a comforting treat.
However, the widespread reports of inconsistent results—ranging from a 50% success rate to achieving perfection only after numerous attempts, or even resorting to adding egg whites—underscore the challenges inherent in its preparation. These failures often prompt cooks to attribute success or failure to seemingly minor, often irrelevant, procedural variations, inadvertently giving rise to "kitchen myths." These anecdotal beliefs, propagated through generations and online forums, frequently offer contradictory instructions, from specific pouring heights to precise stirring regimens, many of which are scientifically unfounded. The quest for a reliable method has driven both amateur cooks and professional food scientists to delve deeper into the molecular gastronomy of this beloved dessert.
A Chronology of Culinary Frustration and Scientific Discovery

The journey from empirical trial-and-error to a scientifically sound understanding of ginger milk curd reflects a broader trend in modern cooking: the integration of culinary arts with scientific principles. Initially, many cooks, including seasoned chefs, approached the dish with a mix of intuition and inherited wisdom. The seemingly random nature of success and failure prompted a deeper investigation.
The first step in unraveling the mystery involved systematically cataloging the diverse range of existing recipes. This revealed a striking lack of consensus on critical parameters such as milk temperature, ginger-to-milk ratio, and mixing techniques. While some recipes advocated for high heat, others warned against it; some insisted on specific ginger varieties, while others focused on the method of juicing. This observational phase underscored the urgent need for a more rigorous, evidence-based approach.
The turning point came with the application of food science principles, particularly biochemistry. Researchers began to isolate and characterize the active components in ginger responsible for milk coagulation. This scientific inquiry moved beyond mere observation, employing laboratory techniques to identify enzymes, measure their activity under varying conditions, and elucidate their interactions with milk proteins. This chronological progression, from culinary frustration to meticulous scientific investigation, ultimately paved the way for a "fool-proof" recipe grounded in reproducible chemical reactions.
The Scientific Blueprint: How Ginger Transforms Milk into a Gel
At the heart of ginger milk curd’s formation lies a fascinating enzymatic process, analogous to the traditional renneting used in cheesemaking. Milk coagulation, in this context, is orchestrated by specific proteolytic enzymes found in fresh ginger, collectively known as ginger proteases (GP), or more specifically, zingipain (EC 3.4.22.67). These enzymes are the tireless workers responsible for initiating the delicate gel network.
Enzymatic Renneting and the Role of Ginger Proteases:
Proteolytic enzymes are biological catalysts that break down proteins into smaller fragments. In cheesemaking, chymosin (rennin) from animal rennet plays this role, selectively cleaving specific milk proteins to induce curd formation. Similarly, ginger proteases target the primary building blocks of milk gels: casein proteins.

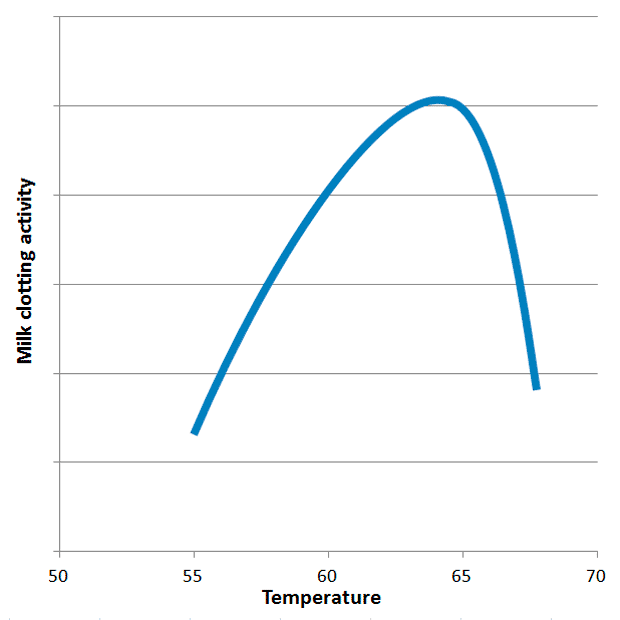
The critical factor governing the success of ginger milk curd is the temperature sensitivity of ginger proteases. Research indicates that GP exhibits peak milk clotting activity (MCA) around 63°C, within a remarkably narrow optimal window of 60-65°C. Outside this range, the enzyme’s efficiency drops sharply. Above 70°C, the proteases are rapidly denatured—irreversibly destroyed—rendering them inactive and preventing gel formation. Below 60°C, their activity is significantly reduced, leading to slow or incomplete coagulation. This precise temperature requirement is the single most important factor explaining the high failure rate observed by many home cooks who do not use a thermometer. For comparison, other plant proteases, such as those from kiwi or melon, have different optimal temperatures (40°C and 70°C, respectively), highlighting the specificity of zingipain.
The Role of Casein Micelles in Gel Formation:
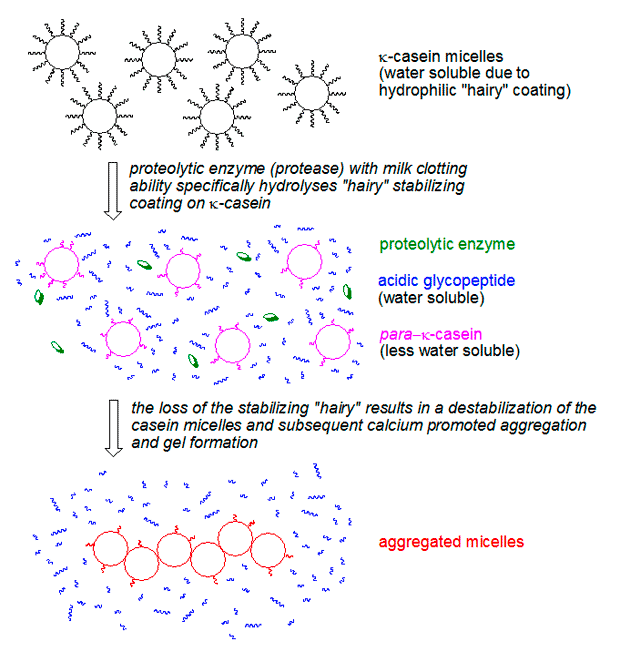
While ginger proteases act as the catalysts, the actual structural components of the gel are casein proteins, which constitute about 80% of milk’s protein content. In liquid milk, casein proteins are organized into complex spherical structures called micelles, stabilized by calcium ions. The outer surface of these micelles is covered by a hydrophilic (water-loving) protein called κ-casein. This κ-casein forms a "hairy layer" that keeps the micelles dispersed in the aqueous phase of milk, preventing them from aggregating and precipitating. This stability is why milk typically remains a liquid.
The magic happens when ginger proteases enter the scene. Zingipain specifically cleaves a peptide bond within the κ-casein molecule, removing its hydrophilic portion. This leaves behind a hydrophobic (water-fearing) fragment called para-κ-casein on the micelle surface. With the protective "hairy layer" gone, the micelles lose their stability and become prone to aggregation. The abundant calcium ions naturally present in milk then act as bridges, facilitating the rapid formation of a three-dimensional network of these "shaved" micelles. This network is the tender, fragile gel we know as ginger milk curd. The entire process occurs within a few minutes, provided the temperature conditions are optimal.
The resulting gel is inherently delicate and prone to syneresis, the process where the gel matrix contracts and expels liquid (whey). This is a natural characteristic of many enzyme-induced protein gels and is visually evident as clear drops of liquid forming around the curd, as shown in the accompanying imagery.
Optimizing the Recipe: Precision for Consistent Success
Armed with this scientific understanding, achieving a "fool-proof" ginger milk curd becomes less about luck and more about precise execution. The following considerations are paramount for consistent success:

1. Temperature Control: This is the most critical parameter. The milk must be heated carefully to 60-65°C. A digital kitchen thermometer is an indispensable tool for this purpose. Heating the milk above 70°C will denature the ginger proteases, leading to failure. Conversely, milk that is too cool will not allow the enzymes to function efficiently. Furthermore, research indicates that heating milk above 65°C (even if cooled afterward) can reduce the strength of the final gel. This is because heat causes other milk proteins, like lactoglobulins, to precipitate onto the κ-casein, interfering with the subsequent enzymatic cleavage and micelle aggregation.
2. Milk Selection: While whole milk can be used, skimmed milk is often recommended for a stronger, more stable gel. Milk fat can interfere with the formation of the casein network, leading to a weaker curd. Similarly, the calcium content of the milk plays a role; a higher calcium concentration can contribute to a stronger gel by enhancing the aggregation of the para-κ-casein micelles.
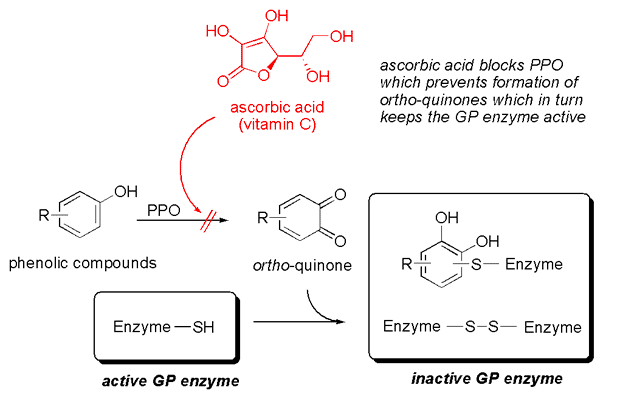
3. Fresh Ginger Juice and Enzyme Stability: The ginger juice itself requires careful handling. Ginger proteases are not indefinitely stable. Freshly squeezed ginger juice has a relatively short half-life of approximately 20 minutes at 30°C. This means that if left at room temperature, half of the enzyme activity will be lost within 20 minutes, and only 25% will remain after 40 minutes. This rapid degradation is due to the presence of another enzyme in ginger, polyphenol oxidase (PPO), the same enzyme responsible for the browning of apples. When ginger is grated, PPO is exposed to air and reacts with phenolic compounds, producing ortho-quinones that can inactivate the ginger proteases.
To circumvent this instability, a simple scientific trick can be employed: adding a small amount of ascorbic acid (Vitamin C), typically around 0.2% by weight, to the ginger juice. Ascorbic acid acts as an antioxidant, inhibiting PPO activity and thereby preserving the integrity and activity of the ginger proteases. This stabilization technique allows ginger juice to be prepared in advance if necessary, although fresh preparation remains ideal for optimal activity.
4. Mixing Technique: Once the milk reaches the precise temperature and the ginger juice is ready, the mixing method is crucial. The milk should be poured into the ginger juice from a moderate height, allowing for rapid and thorough initial mixing without the need for stirring. Stirring after the initial pour can disrupt the nascent gel network as it forms, leading to an inconsistent or failed curd. The mixture should then be left undisturbed at room temperature for 5-10 minutes to allow the gel to set.
Fool-Proof Ginger Milk Curd Recipe:
-
Ingredients:
- 250 mL skimmed milk
- 18 g fresh ginger juice (approximately 31 g peeled ginger or 43 g raw ginger)
- 20 g sugar
-
Equipment:
- Digital kitchen thermometer
- Microplane or grater
- Fine-mesh sieve or cheesecloth for squeezing ginger juice
-
Instructions:
- Combine milk and sugar in a saucepan. Heat carefully over medium-low heat, stirring occasionally, until the temperature reaches precisely 65°C. Do not exceed 65°C. Remove from heat immediately.
- While the milk is heating, peel and microplane or finely grate the fresh ginger. Squeeze out the juice using a fine-mesh sieve or cheesecloth into a serving bowl. (If preparing in advance, add a pinch of ascorbic acid to the juice.)
- Once the milk reaches 65°C, immediately pour it from a moderate height (e.g., 10-15 cm) into the bowl containing the ginger juice. This ensures sufficient initial mixing.
- Do NOT stir after pouring.
- Leave the bowl undisturbed at room temperature. A tender gel will form within 5-10 minutes.
- The ginger milk curd can be served immediately while warm or chilled in the refrigerator.
This recipe uses a milk-to-ginger juice ratio of approximately 14:1. Adjustments can be made for personal taste preferences regarding ginger intensity, but significant reductions in ginger juice may affect gel strength.
Broader Implications: From Kitchen to Industry and Beyond
The scientific elucidation of ginger milk curd’s formation has implications far beyond the home kitchen. It serves as a compelling case study in the power of molecular gastronomy and food science to demystify traditional recipes and elevate culinary practices.
1. Culinary Education and Scientific Literacy: This research underscores the importance of scientific literacy in cooking. Understanding the underlying chemistry and biology of ingredients transforms cooking from a rote following of instructions into an informed, creative process. It empowers cooks to troubleshoot, adapt, and innovate with confidence. Food science curricula increasingly incorporate such examples to illustrate fundamental principles of enzyme kinetics, protein chemistry, and rheology.
2. Food Industry Applications: The knowledge gained from studying ginger proteases has significant industrial relevance.
- Plant-Based Rennet Alternatives: With growing demand for vegetarian and vegan products, plant-derived proteases like zingipain offer viable alternatives to traditional animal rennet for cheese and dairy-free curd production. This opens avenues for developing new lines of specialty cheeses and cultured products.
- Texture and Stability in Food Products: Understanding how enzymes modify protein structures can lead to the development of new food textures and improved product stability. This includes optimizing gelling agents for various dairy and plant-based desserts, as well as understanding how to prevent unwanted protein degradation in other food systems.
- Enzyme Technology: The insights into GP’s temperature sensitivity and stability, particularly the role of PPO and ascorbic acid, contribute to the broader field of enzyme technology, informing methods for enzyme extraction, purification, and stabilization for industrial applications.
3. Innovation in Ingredient Utilization: This research encourages further exploration of natural ingredients as sources of functional enzymes. The success with ginger milk curd prompts questions about other plant extracts and their potential to act as gelling agents or texture modifiers in novel food applications.
4. Consumer Empowerment: By dispelling myths and providing clear, scientifically validated instructions, consumers are empowered to achieve consistent results, reducing food waste from failed attempts and fostering greater enjoyment in cooking. Culinary experts and food scientists alike emphasize that precision in parameters like temperature, ingredient ratios, and preparation techniques is not merely a suggestion but a requirement for achieving reproducible, high-quality outcomes.
Future Avenues for Exploration
While a fool-proof recipe has been established, the scientific journey continues. Several areas warrant further experimentation and research:
- Ginger Variety and Potency: Investigate if different ginger varieties (e.g., old vs. young ginger, specific cultivars) exhibit varying levels of protease activity, potentially influencing gel strength and setting time.
- Optimal pH: Explore the effect of pH on ginger protease activity and gel formation. While milk’s natural pH is relatively stable, slight modifications could potentially optimize the process or allow for the use of other milk types.
- Sugar Concentration: Examine how varying sugar levels impact gel texture and stability, beyond just taste. Sugar can affect protein hydration and aggregation dynamics.
- Milk Alternatives: Test the applicability of this method to plant-based milks (e.g., soy milk, almond milk, oat milk), considering their different protein profiles and potential needs for enzyme optimization or additional gelling agents.
- Scaling Up: Investigate the challenges and modifications required for preparing larger batches of ginger milk curd, where heat retention and mixing dynamics might differ.
The journey from a mysterious, often elusive dessert to a scientifically understood and consistently reproducible culinary delight exemplifies the profound impact of food science. By embracing precision and challenging anecdotal wisdom, we unlock new possibilities in the kitchen, transforming traditionally fickle recipes into reliable culinary triumphs and fostering a deeper appreciation for the intricate chemistry that defines our food.