Beyond the familiar ritual and comforting warmth, a cup of black coffee is a marvel of chemical engineering, a complex aqueous solution composed of hundreds, if not thousands, of distinct molecules that orchestrate its characteristic color, taste, and aroma. While seemingly simple, with over 98.5% of its volume being water, the remaining 1.2-1.5% of total dissolved solids (TDS) – equating to 12-15 grams of extracted compounds per liter of brew – constitutes a microscopic world of profound sensory impact. This intricate composition, painstakingly extracted from approximately 60 grams of roasted coffee grounds, represents a frontier of ongoing scientific discovery and a testament to nature’s nuanced chemistry.
The Foundation of Flavor: Understanding Coffee Extraction Parameters
The journey from bean to brew involves a delicate interplay of various parameters, each significantly influencing the final chemical profile of the coffee. Prior discussions on coffee preparation often highlight factors such as brew ratio, grind size, water composition, water temperature, and filter material. These elements are not merely aesthetic choices but critical levers that dictate which specific compounds, and in what concentrations, are ultimately transferred from the solid coffee matrix into the liquid beverage. For instance, grind size affects the surface area available for extraction; finer grinds expose more surface area, facilitating quicker and more extensive extraction. Water temperature plays a crucial role in solvent efficiency, with hotter water generally dissolving more compounds at a faster rate. Similarly, water composition, including its mineral content, can chemically interact with coffee compounds, influencing both extraction efficiency and the resulting flavor perception. The chosen filter material, whether paper, cloth, or metal, can selectively retain certain insoluble particles and oils, further shaping the coffee’s body and clarity. Mastering these variables allows brewers, from home enthusiasts to professional baristas, to navigate the vast landscape of coffee’s potential, even before delving into its molecular intricacies.
Unveiling the Molecular Tapestry: Analytical Techniques and Unknown Odorants
The identification and characterization of the myriad compounds within coffee are a monumental task, primarily accomplished through advanced chromatographic techniques such as Gas Chromatography-Mass Spectrometry (GC-MS) and High-Performance Liquid Chromatography (HPLC). These sophisticated analytical methods are capable of separating individual chemical components, allowing scientists to pinpoint and often quantify the vast array of molecules present. Despite decades of research, the full spectrum remains elusive. A significant study from 2002, employing aroma extract dilution analysis (AEDA), revealed a striking fact: 13 out of the 40 key odorants contributing to coffee’s distinct aroma remained unidentified. This persistent mystery underscores the complexity of coffee chemistry, where many influential molecules exist in incredibly low concentrations, making their isolation and structural elucidation exceedingly difficult. The sheer size and structural complexity of some molecules also pose analytical challenges, further complicating efforts to resolve their precise chemical makeup.
The difficulty in fully replicating coffee’s natural complexity is further illustrated by early attempts at synthetic "model" coffees. A 1996 study, for instance, managed to create two model coffees, designed to mimic Arabica and Robusta varieties, using a blend of just 22 key volatile compounds. While these synthetic brews were described by assessors as "clearly coffee-like," they conspicuously lacked the full, nuanced aromatic profile of their natural counterparts. This suggests that the 22 compounds, while significant, represent only a fraction of the necessary components to perfectly capture the essence of coffee, highlighting the synergistic effects of numerous trace compounds that collectively contribute to the beverage’s unparalleled richness and depth. The ambition to create a "synthetic" coffee that truly rivals the natural brew remains a distant prospect, affirming the unique and intricate biochemical processes inherent in coffee bean development and roasting.
Volatile vs. Non-Volatile: The Dual Nature of Coffee Sensation
The sensory experience of coffee is bifurcated into two primary categories of compounds: volatile and non-volatile. Volatile compounds, characterized by their low boiling points and readiness to evaporate, are the architects of coffee’s aroma. These molecules waft into the nasal passages, engaging olfactory receptors and creating the complex bouquet that coffee lovers cherish. In contrast, non-volatile compounds remain in the liquid phase and are primarily responsible for the tastes perceived on the tongue, as well as the coffee’s body and texture.

The non-volatile fraction of coffee encompasses a diverse range of chemical groups. Carbohydrates, including various sugars and soluble oligosaccharides, contribute sweetness. A spectrum of acids, such as chlorogenic acids (which break down into quinic and caffeic acids during roasting), citric acid, and acetic acid, provide acidity, brightness, and sometimes bitterness. Minerals and salts play a role in overall flavor balance and mouthfeel. Proteins and lipids (oils) contribute significantly to the coffee’s body and texture, often imparting a creamy or viscous sensation. Caffeine, a well-known alkaloid, is a primary source of bitterness and stimulant properties. Beyond these identified groups, a substantial "unidentified" chunk of compounds exists, many of which are believed to contribute to the coffee’s characteristic color and lingering bitterness. While some molecules can straddle both categories, influencing both aroma and taste, their impact thresholds typically differ, with odor detection often occurring at much lower concentrations than taste perception.
The Power of Trace Compounds: Odor Activity Value and Beta-Damascenone
Perhaps one of the most remarkable aspects of coffee chemistry is the disproportionate impact of certain trace compounds. This phenomenon is best understood through the concept of Odor Activity Value (OAV), defined as the concentration of a compound divided by its specific odor threshold. A high OAV indicates a potent odorant, capable of significantly influencing overall aroma even at exceedingly low concentrations. This explains why a compound present at 0.0000000001% can be as critical to the overall flavor profile as another compound present at 0.01%.
A prime example of such a potent odorant is beta-damascenone. Present in coffee at concentrations as low as 1 part per trillion (ppt), it is nonetheless identified as one of the top five most impactful odorants. Beta-damascenone belongs to a family of molecules known as rose ketones, first isolated from rose oil in the 1960s. Its name is derived from the Damask rose (Rosa × damascena), a flower renowned for its intoxicating fragrance. The flavor and fragrance company Firmenich was instrumental in recognizing the commercial potential of rose ketones, integrating beta-damascenone and its relatives into iconic perfumes such as Dior’s Poison, where they contributed significantly to its distinctive character. The sensory descriptors for beta-damascenone are remarkably complex and varied, including notes of fruity (apple, blackcurrant, plum), floral (rose), woody, tobacco, and honey. This multifaceted profile, detectable at infinitesimal levels, underscores its profound contribution to coffee’s aromatic depth. The precise measurement of odor thresholds and the quantification of compounds at such minute concentrations are incredibly challenging, suggesting that future research may yet uncover additional key odorants with similarly powerful impacts, further enriching our understanding of coffee’s hidden complexities.
Polar vs. Non-Polar Compounds: Decoding Extraction Efficiency
The efficiency with which different compounds are extracted from coffee grounds by water is fundamentally governed by their polarity. Water, being a highly polar solvent, preferentially dissolves and extracts polar compounds. This group includes acids, minerals, proteins, and carbohydrates, which possess molecular structures that readily interact with water molecules. Consequently, more than 75% of these polar compounds are typically extracted during the brewing process.
In contrast, non-polar compounds, such as many volatile aroma compounds and lipids (oils), exhibit low solubility in water. Their molecular structures do not readily form bonds with water molecules, leading to significantly lower extraction yields. Typically, only 10-30% of these non-polar compounds are extracted when coffee is brewed with water. This differential extraction rate has profound implications for brewing methodologies. For instance, longer extraction times or different brewing temperatures might attempt to coax out more of these less soluble, yet often highly aromatic, non-polar compounds. The inherent limitations of water as a solvent for non-polar compounds also opens up avenues for experimental exploration. The use of alternative, less polar solvents, such as ethanol, could potentially unlock entirely new dimensions of coffee extraction, yielding beverages with novel flavor profiles by accessing compounds that largely remain trapped in the coffee grounds when only water is used.
The Limitations of TDS and the Primacy of Human Perception
While Total Dissolved Solids (TDS) serves as a convenient and widely used metric for assessing coffee strength, its utility in predicting the full aroma and flavor profile of a cup of coffee is notably limited. TDS measures the total mass of dissolved solids, primarily reflecting the concentration of the most abundant compounds, such as carbohydrates, acids, and minerals. However, as the logarithmic scale of compound concentrations illustrates, many of the most impactful aroma compounds exist at concentrations several orders of magnitude lower than those that significantly influence TDS readings.
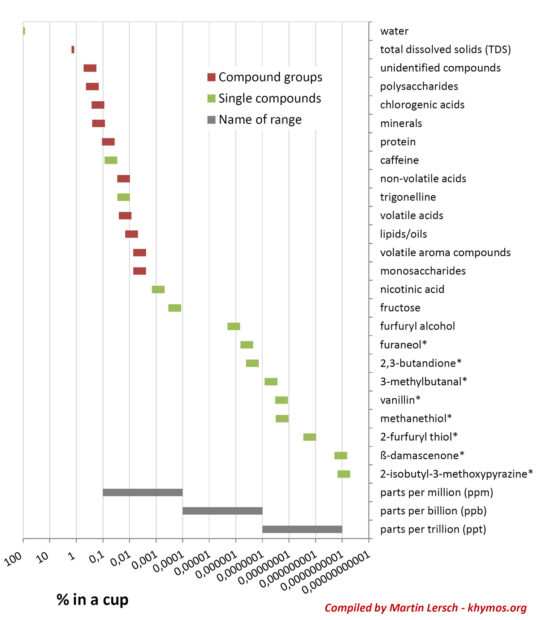
The fundamental flaw in relying solely on TDS for flavor prediction lies in the assumption that all compounds, regardless of their concentration, are extracted at a similar rate, or that concentration directly correlates with sensory impact. This assumption is demonstrably false, as evidenced by the vast difference in extraction yields between polar and non-polar compounds, and the concept of OAV. A high TDS might indicate a strong brew, but it provides little to no information about the balance of delicate floral notes, vibrant fruitiness, or nuanced spice aromas contributed by trace volatile compounds.
Therefore, despite the advent of sophisticated analytical instruments, the human nose and palate remain the most superb and indispensable analytical detectors for evaluating coffee quality. The nuanced interaction of hundreds of compounds, each contributing to a complex sensory mosaic, is something that current scientific instrumentation can quantify but not fully interpret in terms of holistic perception. This is why cupping protocols, sensory panels, and the subjective expertise of tasters continue to be central to the specialty coffee industry, driving innovation and shaping consumer preferences.
Broader Impact and Future Directions
The ongoing scientific exploration into coffee’s molecular composition holds significant implications for the specialty coffee industry, researchers, and consumers alike. For roasters, a deeper understanding of how specific compounds develop or degrade during roasting can lead to more precise profiles that enhance desired flavors and mitigate undesirable ones. Baristas can leverage this knowledge to fine-tune brewing parameters, optimizing extraction to highlight specific aromatic and taste notes. Consumers, in turn, gain a richer appreciation for the complexity in their daily cup, moving beyond simple preferences to a more informed sensory experience.
The scientific community continues to push the boundaries of discovery, with ongoing research focused on identifying more of the "unknown" key odorants and understanding the synergistic effects between compounds. This includes exploring novel extraction methods, analyzing the chemical evolution of coffee from farm to cup, and developing more sophisticated sensory analysis techniques. The quest to fully decipher the wonders of coffee extraction is a continuous journey, ensuring that each cup, despite its apparent simplicity, remains an invitation to explore an unseen universe of flavor and aroma, a testament to the intricate dance of molecules that defines one of the world’s most beloved beverages.