Ginger milk curd, known in Cantonese as 姜汁撞奶 (ginger juice milk collision) or 薑汁燉奶 (ginger juice stewed milk), stands as a testament to culinary ingenuity, a deceptively simple dessert crafted from just three core ingredients: milk, ginger, and sugar. This traditional sweet, cherished for its silken texture and warming ginger spice, presents a fascinating paradox to both home cooks and culinary scientists alike. While its preparation appears straightforward, achieving the perfect tender gel consistently has long been a source of frustration, leading to a proliferation of contradictory advice and widespread "kitchen myths" that obscure its underlying scientific principles.
The Allure of Three Ingredients: A Culinary Paradox
The magic of ginger milk curd lies in its rapid transformation from liquid to a delicate gel within minutes, without the addition of any conventional gelling agents like gelatin or agar. This quick setting, coupled with the inherent appeal of its creamy texture and pungent ginger kick, has made it a beloved treat across various cultures, particularly in East Asia. However, the path to a flawless curd is often fraught with inconsistency. Many enthusiasts report highly variable success rates, with some succeeding only half the time, and others recounting arduous journeys involving numerous failed attempts before achieving the desired consistency. Such experiences underscore the delicate balance of factors at play in this unique enzymatic process.
The journey to demystifying ginger milk curd often begins with personal culinary experimentation. Early attempts by researchers and home cooks alike often yielded surprising successes, quickly followed by perplexing failures despite seemingly identical preparations. This erratic behavior prompted a deeper investigation, moving beyond anecdotal recipes to the rigorous examination of scientific literature. What emerged from this inquiry was a clear understanding that many widely circulated instructions were not only unscientific but actively detrimental to successful curd formation, perpetuating a cycle of trial and error.
Unraveling the Mystery: From Kitchen Fails to Scientific Breakthroughs

The exploration into the scientific basis of ginger milk curd gained significant traction within the broader field of food science, particularly in the context of understanding natural gel formation mechanisms. For instance, research conducted in preparation for comprehensive texts on food texture highlighted ginger milk curd as a prime example of enzyme-driven coagulation in the absence of external hydrocolloids. This academic pursuit brought to light the sheer volume of disparate and often conflicting advice surrounding the dessert’s preparation.
Examples of the conflicting guidance frequently encountered by aspiring ginger milk curd makers include:
- Milk Temperature: Advice ranges wildly, from "heat milk until just steaming" to "bring to a boil and cool slightly," or specific temperatures like "80°C" or "60-70°C." Some recipes even suggest heating milk for an extended period.
- Ginger Preparation: Instructions vary from using "old ginger" to "young ginger," or emphasizing the need to "grate finely" versus "crushing." The quantity of ginger juice is also subject to wide interpretation.
- Mixing Technique: The most contentious area, with some advocating "stirring vigorously," others "stirring gently," and a significant number insisting on "no stirring at all" after pouring. The height from which milk is poured is also a point of debate.
- Setting Conditions: Recommendations include "setting in a warm place," "setting in the fridge," or "setting at room temperature."
This spectrum of advice, often presented without scientific justification, creates a fertile ground for "kitchen myths." These myths arise when individuals, experiencing random successes and failures, attempt to correlate their last action with the outcome. A successful batch might lead to the erroneous conclusion that a specific, perhaps irrelevant, alteration was the key, solidifying a myth that then gets passed down. The prevalence of such myths explains why reports of success rates as low as 50% or requiring nine attempts are common, sometimes leading cooks to abandon the traditional method altogether in favor of adding egg whites for guaranteed setting.
Precision in the Pan: The Fool-Proof Ginger Milk Curd Recipe
Based on rigorous scientific understanding, a reliable method for preparing ginger milk curd emphasizes precision, particularly regarding temperature and mixing. The most crucial tool for consistent success is a digital kitchen thermometer, allowing for accurate temperature control—a factor consistently overlooked in traditional, myth-laden recipes.

Fool-proof Ginger Milk Curd Recipe:
- 250 mL skimmed milk
- 18 g fresh ginger juice (approximately 31 g peeled ginger or 43 g raw ginger)
- 20 g sugar
Instructions:
- Prepare Milk: Combine milk and sugar in a saucepan. Heat carefully, stirring occasionally, until the mixture reaches precisely 65°C. Remove immediately from heat.
- Prepare Ginger Juice: While the milk is heating, peel fresh ginger and microplane it to maximize surface area for juice extraction. Firmly squeeze out 18 grams of fresh ginger juice into a serving bowl. Note: Ginger juice should be prepared immediately before use due to enzyme instability.
- Combine and Set: Once the milk reaches 65°C, pour it from a moderate height (about 15-20 cm) directly into the bowl containing the ginger juice. This pouring technique ensures sufficient initial mixing without the need for stirring.
- Crucial Step – Do NOT Stir: After pouring, do not stir the mixture. Stirring can disrupt the delicate network of proteins forming the gel.
- Allow to Set: Leave the bowl undisturbed at room temperature. A tender gel will typically form within 5-10 minutes. The curd can be served immediately, ideally warm, or chilled in the refrigerator for later consumption.
This recipe utilizes a milk-to-ginger juice ratio of approximately 14:1. While this ratio provides a balanced ginger flavor and reliable set, variations in ginger concentration can be explored for different taste preferences, though significantly reducing ginger juice may impact gelling efficiency.
The Enzymatic Heart: How Ginger Transforms Milk
The remarkable transformation of milk into a gel by ginger is a classic example of enzyme-catalyzed coagulation, a process fundamental to many food preparations, including cheesemaking. Just as rennet, derived from the stomachs of young mammals, uses the enzyme chymosin (rennin) to curdle milk, ginger relies on its own proteolytic enzymes.

Proteases at Play: Ginger contains potent proteases, collectively known as ginger proteases (GP), with one well-characterized enzyme being zingipain (EC 3.4.22.67). These enzymes are specialists in proteolysis – breaking down proteins into smaller fragments. In the context of milk, their primary target is casein.
Temperature: The Goldilocks Zone: The activity of ginger proteases is exquisitely sensitive to temperature. Research indicates that their milk clotting activity (MCA) peaks sharply around 63°C. The effective temperature window is remarkably narrow, spanning roughly 60-65°C. Above 70°C, these delicate enzymes rapidly denature, meaning their three-dimensional structure is irreversibly destroyed, rendering them inactive. Below 60°C, while some non-specific proteolytic activity may persist, the specific MCA required for efficient curdling diminishes significantly. This critical temperature dependence is the single most common reason for failure in ginger milk curd preparation and underscores the necessity of a thermometer. Other plant proteases, such as those found in kiwi (optimal at 40°C) and melon (optimal at 70°C), exhibit different temperature profiles for their milk-clotting capabilities, highlighting the unique properties of ginger’s enzymes.
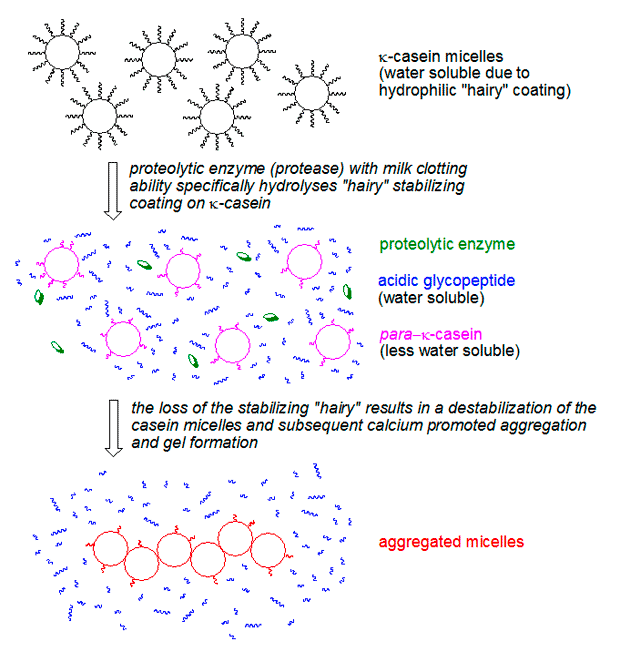
Casein: The Building Blocks of the Gel: Milk’s primary proteins, casein, are the structural components of the gel. These proteins aggregate into large colloidal structures called micelles, stabilized by calcium ions. Crucially, the surface of these micelles is coated with a "hairy layer" of kappa-casein. This layer, composed of a water-soluble glycopeptide portion and a less soluble para-kappa-casein portion, acts as a protective barrier. It keeps the micelles dispersed in the aqueous phase of milk, preventing them from aggregating and precipitating. In essence, this "hairy layer" is what makes milk a stable liquid suspension rather than a spontaneous gel.
The Transformation: When ginger proteases are introduced to milk at the optimal temperature, they act specifically on the kappa-casein. The enzymes cleave off the water-soluble glycopeptide part, leaving behind the hydrophobic para-kappa-casein. With their protective "hairy layers" removed, the now "shaved" casein micelles lose their colloidal stability. They begin to collide, aggregate, and form a three-dimensional network, a process significantly aided by the calcium naturally present in milk. This rapid aggregation is what forms the tender, fragile gel within minutes.
Syneresis Explained: The resulting gel is inherently delicate and prone to syneresis, the process where a gel expels liquid (in this case, whey). This occurs because the protein network contracts over time, squeezing out the trapped water molecules. The clear liquid often seen collecting at the bottom of a ginger milk curd bowl, or as a drop under a spoon, is a direct visual manifestation of syneresis.
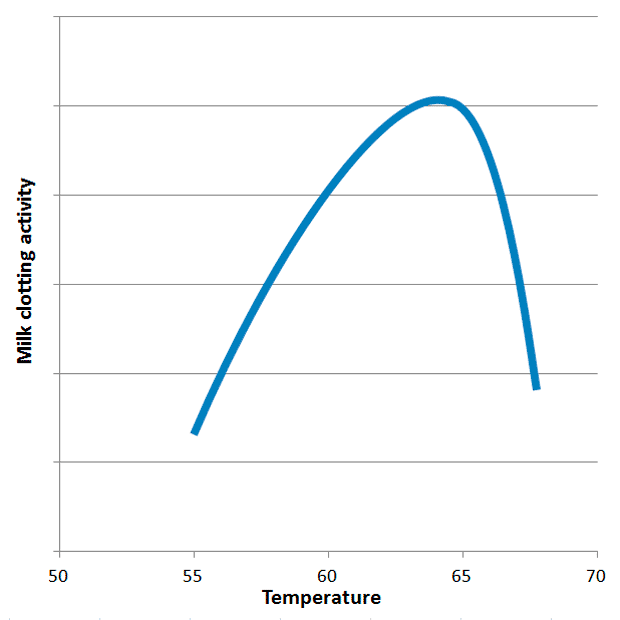
Factors Influencing Gel Strength and Stability
Beyond the optimal temperature for enzymatic activity, several other factors critically influence the strength and stability of the ginger milk curd.
Milk Composition:
- Fat Content: Skimmed milk is generally preferred for a stronger gel. Milk fat can interfere with the formation of the protein network, leading to a weaker, less cohesive curd.
- Heat Treatment of Milk: Heating milk to temperatures above 65°C before cooling it to the optimal range can paradoxically weaken the final gel. This is because high heat causes other milk proteins, particularly lactoglobulins, to denature and precipitate onto the kappa-casein. This coating then obstructs the ginger proteases from effectively cleaving the kappa-casein, hindering proper gel formation.
- Calcium Concentration: Since calcium ions play a crucial role in bridging and aggregating the "shaved" casein micelles, a higher calcium concentration in milk generally leads to a stronger gel.
Ginger Juice: A Race Against Time:
The ginger juice itself requires careful handling due to the inherent instability of its proteases. Freshly squeezed ginger juice, if left at room temperature (e.g., 30°C), experiences a rapid decline in protease activity. Ginger proteases have a half-life of approximately 20 minutes under these conditions. This means that after 20 minutes, half of the enzyme’s activity is lost, and after another 20 minutes, only 25% of the original activity remains. This rapid degradation explains why preparing ginger juice in advance or storing it at room temperature often leads to failed curd attempts.
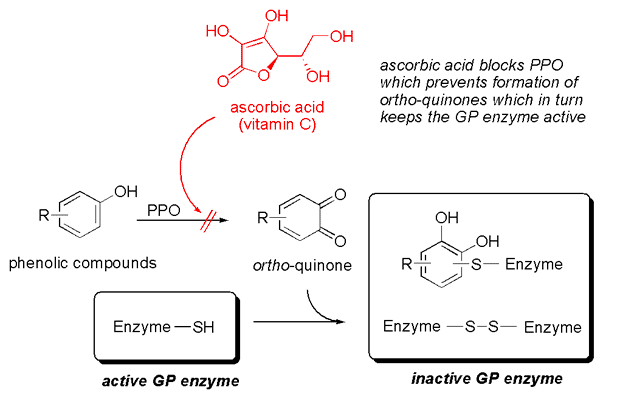
The culprit behind this instability is another enzyme present in ginger: polyphenol oxidase (PPO). PPO is responsible for the enzymatic browning seen in fruits like apples. When ginger is grated or cut, PPO is exposed to air and phenolic compounds, initiating a reaction that produces ortho-quinones. These quinones can then react with and inactivate the ginger proteases, effectively "poisoning" the very enzymes needed for curdling.
The Ascorbic Acid Solution: A simple yet elegant solution to stabilize ginger juice involves the addition of ascorbic acid (Vitamin C). Ascorbic acid acts as an antioxidant, blocking the action of PPO. By inhibiting PPO, it prevents the formation of quinones, thereby protecting the ginger proteases from inactivation. Adding a small pinch (approximately 0.2% by weight) of vitamin C to freshly squeezed ginger juice can significantly extend the stability of the ginger proteases, making it possible to prepare the juice slightly in advance without compromising its gelling power.
Beyond the Kitchen: Broader Implications and Future Directions
The scientific elucidation of ginger milk curd’s mechanism has far-reaching implications, extending beyond perfecting a traditional dessert.
Culinary Science and Innovation: For home cooks and professional chefs, this understanding transforms a notoriously fickle recipe into a predictable and reproducible culinary endeavor. It empowers them to move beyond superstition and embrace precision, fostering a deeper appreciation for the chemistry of cooking. This shift aligns with broader trends in culinary science, where scientific principles are applied to innovate and refine food preparation.
Industrial Applications: The study of ginger proteases, alongside other plant-derived enzymes like those from kiwi and melon, holds significant promise for the food industry. These plant-based proteases represent potential alternatives to traditional animal rennet in cheesemaking, catering to vegetarian and vegan markets or addressing concerns about animal welfare. Further research could optimize their activity, specificity, and yield for commercial applications in dairy processing.
Educational Value: Ginger milk curd serves as an excellent, hands-on demonstration for science education, from high school chemistry projects to undergraduate food science labs. It visually illustrates concepts such as enzyme kinetics, protein denaturation, colloidal chemistry, and the impact of environmental factors like temperature on biochemical reactions.
Untapped Potential and Further Experimentation: Despite the significant strides in understanding ginger milk curd, several avenues remain open for further exploration, inviting curious minds to continue the scientific inquiry:
- Investigating the precise impact of different ginger varieties, ages, and preparation methods on protease activity and flavor.
- Exploring the optimal milk-to-ginger juice ratio for various desired gel strengths and textures.
- Analyzing the effect of different sugar types and concentrations on gel formation and stability.
- Delving deeper into the role of milk pH and mineral content (e.g., calcium) in curd integrity.
- Experimenting with alternative natural inhibitors for PPO to stabilize ginger juice.
- Quantifying the effect of various stirring techniques (or lack thereof) more rigorously on the final gel structure using rheological analysis.
Ultimately, the humble ginger milk curd embodies a sophisticated interplay of biochemistry and culinary art. By peeling back the layers of tradition and myth with scientific inquiry, we not only gain a "fool-proof" recipe but also a profound appreciation for the intricate natural processes that shape our food, turning a challenging dessert into a celebration of edible science.