The journey into the seemingly simple realm of ginger milk curd, or 薑汁撞奶/姜汁撞奶 as it is known in Chinese cuisine, reveals a fascinating interplay of culinary art and molecular gastronomy. What begins as an exploration of a dessert requiring just three ingredients – milk, ginger, and sugar – quickly evolves into a deep dive into enzymatic reactions, protein coagulation, and the persistent challenge of kitchen myths. The initial allure of forming a tender, fragile gel within minutes, boasting a robust ginger flavor with a hint of sweetness, often gives way to frustrating inconsistencies for home cooks, highlighting a common disconnect between traditional recipes and underlying scientific principles.
The Culinary Conundrum: Unpacking Kitchen Myths
The quest for a consistently successful ginger milk curd often leads cooks down a winding path of contradictory advice. Anecdotal evidence abounds, with reports ranging from 50% success rates to recipes finally working on the ninth attempt, or even cooks abandoning the core ingredients to introduce egg whites out of sheer desperation. This landscape of hit-or-miss outcomes is fertile ground for the propagation of "kitchen myths." These myths typically emerge when individuals attempt to attribute successful or failed results to perceived changes in their methodology, often without a true understanding of the chemical or physical processes at play. A seemingly minor alteration, or even an unrelated factor, can be mistakenly identified as the critical variable, leading to elaborate and often illogical instructions that lack scientific grounding.
For instance, common advice found across various culinary platforms includes:

- Temperature of the milk: Suggestions range wildly from "boiled hot milk" to "scalding milk" or specific temperatures like "80°C," "75°C," "70°C," or "just below boiling." Some even suggest "any temperature above 60°C" or "milk should be warm, not hot."
- Pouring technique: Some advocate for "pouring from a height" while others insist on "stirring vigorously" or "not stirring at all."
- Ginger juice preparation: Advice varies on whether to use "aged ginger," "young ginger," or a specific amount of "ginger juice," with little guidance on its freshness or stability.
- Setting conditions: Instructions might suggest "covering the bowl" or "leaving it uncovered," or even placing it in specific environments like a "warm spot" or "at room temperature."
This bewildering array of instructions underscores the difficulty cooks face when attempting a recipe that, despite its apparent simplicity, relies on precise biochemical conditions. The author’s own experience, moving from initial success to multiple failures before consulting scientific literature, epitomizes the journey from culinary intuition to evidence-based cooking.
A Scientific Breakthrough: The Fool-Proof Recipe Revealed
Through rigorous investigation, blending culinary experimentation with scientific research, a reliable method for ginger milk curd has been developed. The key to consistency lies in meticulous control over critical variables, primarily temperature and ingredient freshness. The resulting "fool-proof" recipe, based on current scientific understanding, minimizes the guesswork and maximizes the chances of success:
Fool-Proof Ginger Milk Curd Recipe:
- Ingredients:
- 250 mL skimmed milk
- 18 g fresh ginger juice (approximately 31 g peeled ginger or 43 g raw ginger)
- 20 g sugar
- Method:
- Combine skimmed milk and sugar in a saucepan. Heat carefully, monitoring with a digital kitchen thermometer, until the mixture reaches precisely 65°C. This temperature is crucial for optimal enzyme activity.
- While the milk heats, peel fresh ginger and microplane it to maximize juice extraction. Squeeze out 18 grams of fresh ginger juice into a serving bowl.
- Once the milk reaches 65°C, immediately pour it into the ginger juice from a moderate height. This action ensures adequate mixing without the need for stirring, which can disrupt the delicate gel formation.
- Do NOT stir after pouring. Allow the mixture to set undisturbed at room temperature. A stable gel will typically form within 5-10 minutes.
- The curd can be served immediately or chilled for later enjoyment.
This recipe emphasizes a milk-to-ginger juice ratio of approximately 14:1, which provides a balanced ginger flavor without being overpowering. Variations in ginger juice quantity can be explored for personal preference, though reduction may impact gel strength, and significant increase could alter the flavor profile too drastically. A critical note is the absolute necessity of fresh ginger juice, as the enzymes responsible for coagulation are highly unstable.

The Biochemistry of Gelation: Ginger Proteases at Work
The transformation of liquid milk into a solid gel is a remarkable feat of enzymatic action, a process widely recognized in the production of cheese using rennet. Rennet, traditionally sourced from the stomachs of young mammals, contains the enzyme chymosin (also known as rennin), a proteolytic enzyme or protease. These enzymes specialize in breaking down proteins into smaller fragments.
Ginger enters this biochemical narrative because it too harbors powerful proteolytic enzymes, collectively known as ginger proteases (GP), with zingipain (EC 3.4.22.67) being a prominent example. These enzymes are the unsung heroes of ginger milk curd formation, acting as catalysts to initiate milk coagulation. However, their efficacy is highly dependent on environmental conditions, particularly temperature.
The milk clotting activity (MCA) of ginger proteases exhibits a narrow optimal temperature window, peaking sharply around 63°C. Activity drops off rapidly above 65°C, where the enzymes begin to denature irreversibly, and below 60°C, where their catalytic efficiency significantly diminishes. This precise temperature requirement, often overlooked in traditional recipes, is the primary reason for widespread failures in ginger milk curd preparation. While ginger proteases possess general proteolytic activity outside this window, their specific milk-clotting action, which involves the hydrolysis of κ-casein, is largely confined to the 60-65°C range.
The discovery of plant-based proteases with milk-clotting capabilities has significant implications for the dairy industry, offering alternatives to animal-derived rennet, particularly for vegetarian and vegan products. Scientists have identified similar proteases in other fruits like kiwi (optimal MCA at 40°C) and melon (optimal MCA at 70°C), each presenting unique temperature profiles and potential applications in food processing and new product development, including plant-based cheeses.

Casein Micelles: The Building Blocks of the Curd
At the heart of milk coagulation lies casein, a family of phosphoproteins constituting about 80% of the protein in cow’s milk. Casein proteins exist in milk as large, complex structures called micelles, which are suspended in the aqueous phase. These micelles are not simply aggregates but highly organized structures, stabilized by calcium phosphate and a protective outer layer of κ-casein.
The κ-casein molecules on the surface of the micelles are unique: they possess a highly water-soluble segment (an acidic glycopeptide) that extends outwards into the milk serum, creating a "hairy" protective layer. This layer serves two crucial functions: it keeps the casein micelles dispersed and dissolved in water, preventing them from settling out, and it creates steric hindrance and electrostatic repulsion, preventing the micelles from clumping together and aggregating spontaneously. This stability is what keeps milk in its liquid state.
When ginger proteases, chymosin, or other milk-clotting enzymes are introduced to milk, they target this κ-casein layer. Specifically, these proteases cleave the water-soluble part of the κ-casein, leaving behind a less soluble component known as para-κ-casein. This enzymatic cleavage effectively "shaves" the hairy layer off the micelles, removing their protective barrier. Without this barrier, the now exposed hydrophobic surfaces of the para-κ-casein micelles are free to interact. In the presence of calcium ions, which are abundant in milk, these "shaved" micelles rapidly collide, aggregate, and link together to form a three-dimensional protein network – the gel. This process is remarkably fast, typically occurring within minutes. The resulting gel, characterized by its tender and fragile nature, is prone to syneresis – the expulsion of whey (liquid) from the gel matrix, often observed as a clear drop under a spoon.
Factors Influencing Gel Strength and Stability
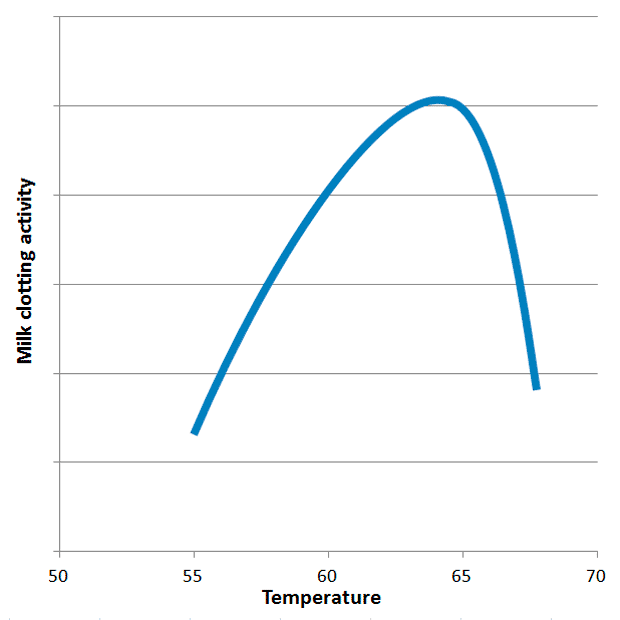
Beyond the critical role of temperature and enzyme activity, several other factors influence the strength and stability of the ginger milk curd:
- Milk Type and Heat Treatment: Heating milk to excessively high temperatures (above 65°C) before cooling it down can inadvertently weaken the final gel. This is because heat causes whey proteins, particularly lactoglobulins, to denature and precipitate onto the κ-casein. This coating on the κ-casein can interfere with the specific cleavage action of ginger proteases, hindering the proper aggregation of micelles and resulting in a softer, less stable gel. For this reason, skimmed milk is often preferred in recipes aiming for a stronger gel, as milk fat can also interfere with protein network formation.
- Calcium Concentration: Calcium ions play a pivotal role in cross-linking the "shaved" casein micelles, facilitating their aggregation into a robust gel network. A higher concentration of calcium can therefore lead to a stronger and firmer curd. This is a common principle utilized in cheesemaking to achieve desired texture profiles.
- Syneresis: The characteristic fragility of ginger milk curd and its tendency to release liquid (whey) is a phenomenon known as syneresis. While often seen as a defect in other food gels, it is inherent to this enzymatic coagulation process. Understanding syneresis is key to appreciating the delicate texture of ginger milk curd and managing its presentation.
The Volatility of Ginger Juice: Enzymes and Oxidation
The effectiveness of ginger juice in forming a curd is not only temperature-dependent but also time-dependent. Ginger proteases are surprisingly unstable once extracted from the ginger root. In freshly squeezed ginger juice, the milk-clotting enzymes have a half-life of only about 20 minutes at 30°C. This means that if ginger juice is prepared in advance and left at room temperature, half of its enzymatic activity will be lost within 20 minutes, and only 25% will remain after 40 minutes. This rapid degradation is another major contributor to recipe failures and the rise of kitchen myths, as cooks might blame other factors when the real issue is simply inactive ginger juice.
The culprit behind this enzymatic instability is another enzyme present in ginger: polyphenol oxidase (PPO). PPO is notorious for causing the enzymatic browning of fruits and vegetables, such as apples, when exposed to air. In ginger, once the tissue is damaged (e.g., by grating), PPO catalyzes the oxidation of phenolic compounds into ortho-quinones. These highly reactive quinones can then irreversibly bind to and inactivate the ginger proteases, rendering them incapable of coagulating milk.
Fortunately, there’s a simple scientific solution to stabilize ginger juice: ascorbic acid, commonly known as Vitamin C. Ascorbic acid acts as an antioxidant and a reducing agent, effectively blocking the action of PPO. By preventing the formation of ortho-quinones, ascorbic acid protects the ginger proteases from inactivation. Adding a small pinch (approximately 0.2%) of Vitamin C to freshly squeezed ginger juice can significantly extend the half-life of the ginger proteases, allowing for the juice to be prepared slightly in advance without losing its potency. This simple trick transforms a volatile ingredient into a more manageable one, further enhancing the reliability of the ginger milk curd recipe.
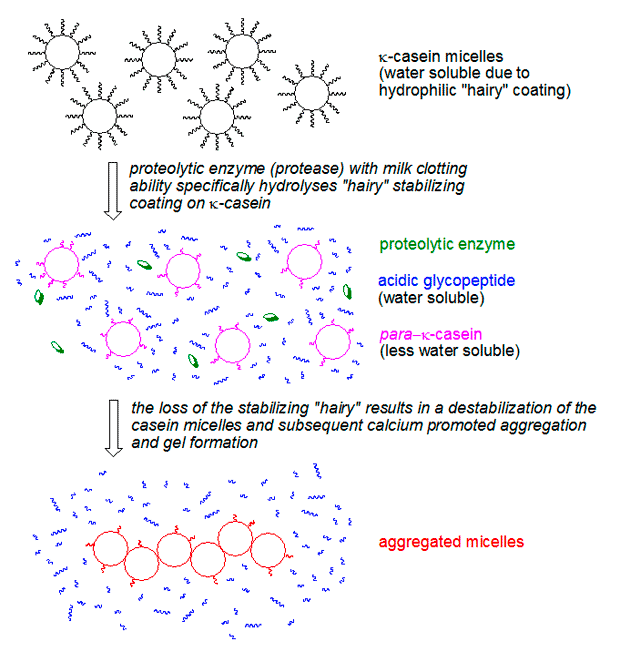
Beyond the Recipe: Implications for Culinary Science and Innovation
The scientific understanding of ginger milk curd extends far beyond a single dessert. It highlights the broader implications of applying scientific principles to culinary practices, demonstrating how a deeper understanding of ingredients and processes can demystify complex preparations and debunk persistent kitchen myths.
For home cooks, this means moving from guesswork to informed cooking, fostering a greater appreciation for the "why" behind successful techniques. The emphasis on precision, such as using a digital thermometer and understanding enzyme kinetics, elevates cooking from a mere art to an accessible form of applied science.
For the food industry and scientific community, the study of ginger milk curd contributes to a growing body of knowledge on plant-derived enzymes and their potential applications. The identification and characterization of plant proteases with milk-clotting activity offer sustainable and ethical alternatives to animal rennet, paving the way for innovations in vegetarian and vegan dairy products. Furthermore, this research deepens our understanding of protein-protein interactions, gel formation, and food texture, areas critical for developing new food products with desired sensory attributes and shelf stability. The meticulous investigation into enzyme stability and the role of anti-browning agents also provides valuable insights for optimizing ingredient preparation and preservation in a wider range of food applications.
The journey from a simple three-ingredient recipe to a comprehensive scientific explanation of ginger milk curd underscores the profound connection between the kitchen and the laboratory. It demonstrates that with careful observation, experimentation, and a commitment to scientific inquiry, even the most elusive culinary successes can be made consistently achievable. The ongoing exploration of such phenomena promises to continue enriching both our understanding of food and our enjoyment of it.