Every morning, millions worldwide partake in a ritual as old as time: brewing a cup of coffee. While the comforting warmth and stimulating effects are universally recognized, the true complexity of this beloved beverage extends far beyond its dark appearance and familiar aroma. Scientifically, a standard cup of black coffee is an astonishingly intricate chemical solution, consisting of over 98.5% water and a mere 1.2-1.5% of total dissolved solids (TDS). This seemingly small fraction, translating to 12-15 grams of extracted compounds per liter of brew from approximately 60 grams of coffee grounds, orchestrates a symphony of hundreds, if not thousands, of distinct molecules that define its color, taste, and aroma.
A Historical Perspective on Coffee Chemistry
The human fascination with coffee’s properties dates back centuries, with early observations focused primarily on its stimulating effects and distinct flavor profile. However, a scientific understanding of its chemical constituents only began to emerge with the advent of sophisticated analytical techniques in the 20th century. Before the widespread adoption of tools like gas chromatography-mass spectrometry (GC-MS) and aroma extract dilution analysis (AEDA), coffee’s flavor was largely a mystery, attributed vaguely to "coffee oils" or "essences."
The 1960s and 70s marked a significant turning point, as chemists started to meticulously separate and identify individual volatile compounds responsible for aroma. This period saw the foundational work that laid the groundwork for modern coffee science. By the 1990s and early 2000s, studies like those by Semmelroch and Grosch (1996) and Sanz et al. (2002) leveraged these advanced techniques to delve deeper into the specific odorants and their relative contributions. The 2002 AEDA study, for instance, delivered a startling revelation: 13 of the 40 key odorants in coffee remained unidentified, underscoring the vast uncharted territory in coffee chemistry even then. This ongoing quest to map coffee’s molecular landscape represents a significant chronology in food science, moving from basic empirical observations to highly precise molecular characterization.
Deconstructing the Brew: Volatiles, Non-Volatiles, and Their Roles
The compounds within a cup of coffee can be broadly categorized into volatile and non-volatile components, each playing a critical, albeit different, role in the overall sensory experience.
The Aroma Ensemble: Volatile Compounds
Volatile compounds are those that readily evaporate, making their way into the olfactory receptors in our nose and defining coffee’s complex aroma. These molecules are primarily formed during the roasting process through intricate chemical reactions like the Maillard reaction (responsible for browning and savory notes) and Strecker degradation. These reactions transform amino acids and sugars into a diverse array of aromatic compounds. Key classes of volatile compounds include:
- Pyrazines: Often contribute roasted, nutty, earthy notes.
- Furans: Impart caramel-like, sweet, and sometimes burnt sugar notes.
- Aldehydes: Can offer fruity, green, or malty aromas.
- Ketones: Contribute a wide range of notes, from fruity to floral.
- Thiols (Sulfur compounds): Present in extremely low concentrations, yet crucial for roasted, meaty, or even rubbery nuances (though often desirable in moderation).
The challenge in identifying all key aroma compounds lies in their often minute concentrations and the sheer number of different molecules present. Some are so dilute that obtaining sufficient quantities for full structural elucidation is difficult, while others possess complex structures that complicate analysis. The fact that synthetic "model" coffees, even those based on 22 key volatile compounds, could only be described as "clearly coffee-like" by assessors, highlights the immense synergistic complexity required to replicate the true coffee aroma profile. This ongoing research by institutions like the Coffee Science Centre and university food chemistry departments continues to seek the complete "fingerprint" of coffee aroma. Dr. Elena Rodriguez, a leading food chemist specializing in flavor science, commented in a recent symposium, "The subtlety of coffee’s aroma is its greatest enigma and its greatest allure. We are still uncovering layers of complexity, proving that even with advanced technology, nature’s recipes are incredibly difficult to fully decode."

The Taste Foundation: Non-Volatile Compounds
Non-volatile compounds, conversely, remain in the liquid and interact with taste receptors on the tongue, contributing to the fundamental tastes of bitterness, sweetness, acidity, and astringency. Beyond taste, non-volatiles, alongside insoluble particles and oils, also contribute to the coffee’s "body" and "mouthfeel."
- Acids: A cornerstone of coffee’s taste profile. Chlorogenic acids are the most abundant, contributing significantly to bitterness and astringency, especially in darker roasts where they break down into quinic and caffeic acids. Other acids like citric, malic, and acetic acids contribute fruity, tart, and bright notes, particularly in lighter roasts and Arabica varieties.
- Carbohydrates: Simple sugars (glucose, fructose, sucrose) contribute sweetness, while soluble oligosaccharides can influence body and mouthfeel.
- Alkaloids: Caffeine is the most well-known, providing the stimulating effect. It also imparts a significant bitter taste, though its perception varies greatly among individuals due to genetic factors. Other minor alkaloids exist but have less impact on flavor.
- Lipids (Oils) and Proteins: While often present in small amounts in filtered coffee, they are crucial for crema formation in espresso and contribute to body and mouthfeel, particularly in French press or unfiltered brews.
- Minerals/Salts: Trace minerals extracted from the bean and present in the brewing water can subtly influence taste and enhance other flavor perceptions.
A significant portion of non-volatile compounds also remain unidentified, many of which likely contribute to the coffee’s characteristic color and overall bitterness.
The Potent Power of the Minute: Beta-Damascenone as a Case Study
One of the most compelling aspects of coffee chemistry is the concept of Odor Activity Value (OAV), defined as a compound’s concentration divided by its odor threshold. This metric explains why a compound present in infinitesimally small quantities can have a monumental impact on overall flavor. Beta-damascenone stands as a prime example of this phenomenon.
Despite being present in coffee at concentrations as low as 1 part per trillion (ppt) – equivalent to one drop of water in 20 Olympic-sized swimming pools – beta-damascenone is consistently identified as one of the top five most impactful odorants in coffee. Its remarkable potency stems from the extraordinary sensitivity of the human nose to this particular molecule. Its aroma profile is complex and highly prized: often described as sweet, fruity (particularly apple, rose, and blackcurrant), floral, and woody, it contributes significantly to the overall bouquet of a high-quality coffee.
Beta-damascenone belongs to a family of compounds known as rose ketones, first discovered in rose oil in the 1960s. Its name is derived from the Damask rose (Rosa × damascena), a flower renowned for its intoxicating fragrance. The Swiss flavor and fragrance company Firmenich was instrumental in pioneering the commercial application of rose ketones, notably incorporating beta-damascenone into the iconic Dior perfume, Poison. This history illustrates how scientific discoveries in one field can profoundly impact others, transforming both the culinary and cosmetic industries. Accurately measuring such minute concentrations and their corresponding odor thresholds remains a challenging, painstaking endeavor for analytical chemists, suggesting that more potent, low-concentration odorants in coffee may yet be discovered.
The Extraction Enigma: Polar vs. Non-Polar Dynamics
The process of brewing coffee is fundamentally an act of selective extraction, governed by the interaction between water and the chemical compounds within the coffee grounds. Water, being a highly polar solvent, preferentially extracts polar compounds. This group includes acids, minerals, proteins, and carbohydrates, making them highly soluble in water. Consequently, typical brewing methods efficiently extract more than 75% of these polar compounds from the coffee grounds.
However, water’s polarity presents a limitation when it comes to non-polar compounds. This category includes many of the volatile aroma compounds and lipids (oils). Their low solubility in water means that conventional brewing techniques typically extract only 10-30% of these crucial non-polar components. This disparity in extraction efficiency between polar and non-polar compounds is a critical factor in understanding the final flavor profile. It also opens avenues for exploration: consider the potential of less polar solvents, such as ethanol, which could selectively extract a different spectrum of compounds, yielding novel flavor dimensions. This concept has implications for innovative beverage development and experimental coffee preparations.
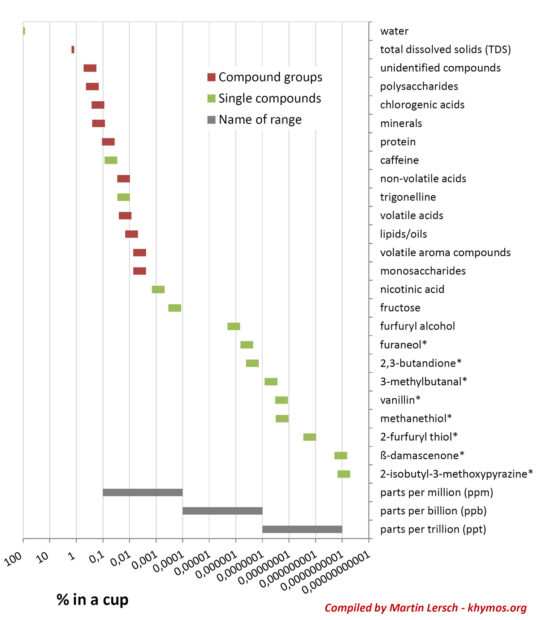
Beyond Total Dissolved Solids: A Call for Comprehensive Analysis
While Total Dissolved Solids (TDS) is a widely used metric in the coffee industry, serving as a reliable indicator of brew strength, its utility as a predictor of aroma and flavor is notably limited. A TDS reading, typically ranging from 1.2% to 1.5% in black coffee, primarily reflects the concentration of the most abundant soluble compounds, such as carbohydrates, acids, and certain minerals. These compounds are present in sufficient quantities to significantly influence the overall mass of dissolved solids.
The inherent flaw in relying solely on TDS for flavor assessment lies in the fact that it does not account for the differential extraction rates of various compound groups, nor does it capture the impact of highly potent, low-concentration aroma molecules. As previously discussed, polar compounds are efficiently extracted, contributing substantially to TDS, while many critical non-polar aroma compounds are extracted much less efficiently. Furthermore, a compound like beta-damascenone, present in parts per trillion, will have virtually no impact on a TDS measurement, yet it can be a dominant force in the perceived flavor.
Therefore, a TDS measurement, while useful for consistency in strength, is a very poor predictor of the nuanced aroma and complex flavor profile in a cup of coffee. The scientific community and specialty coffee professionals increasingly acknowledge that a holistic understanding requires moving beyond simple metrics. This necessitates a multi-faceted approach, combining advanced analytical chemistry with sensory evaluation. The human nose, with its unparalleled sensitivity and ability to integrate a myriad of signals, remains the most superb and sophisticated analytical detector for the intricate wonders of coffee extraction.
Industry Insights and Future Horizons
The deepening scientific understanding of coffee’s chemical composition carries profound implications for the global coffee industry, from bean cultivation to brewing innovation. Specialty coffee roasters and baristas are increasingly leveraging this knowledge to refine their processes, optimize roasting profiles, and develop precise brewing parameters that highlight specific flavor notes. "Understanding the molecular basis of flavor allows us to move beyond intuition to truly craft and control the sensory experience," states Dr. Marcus Thorne, a research lead at a major coffee corporation. "It’s about consistency, quality, and unlocking new potential in every bean."
This research also fuels advancements in flavor science, with implications for synthetic food and beverage development. While perfectly replicating coffee’s complexity remains a formidable challenge, the identification of key odorants and their thresholds paves the way for more sophisticated flavor formulations in other food products. Looking ahead, the integration of artificial intelligence and machine learning with advanced analytical techniques promises to accelerate the discovery of unknown compounds and predict optimal extraction conditions. Further research into health-beneficial compounds beyond caffeine, such as various antioxidants and polyphenols, is also a rapidly expanding field, adding another dimension to coffee’s allure.
Conclusion: The Enduring Mystery and Allure
From a deceptively simple liquid, coffee reveals itself as a marvel of chemical engineering, a complex interplay of hundreds of molecules working in concert. The journey from humble bean to fragrant brew is a testament to both natural processes and meticulous human intervention. While scientists continue their tireless quest to map every molecule and unravel every interaction, the enduring mystery of coffee’s full sensory impact only deepens its allure. Ultimately, the art of brewing and the science of its composition converge in that daily ritual, offering a profound sensory experience that continues to captivate and inspire.