The unassuming cup of black coffee, a ubiquitous daily ritual for billions, belies a profound chemical complexity that continues to fascinate scientists and connoisseurs alike. Far from a simple beverage, it is a meticulously crafted extraction, primarily composed of water yet containing a dazzling array of dissolved solids that orchestrate its distinctive color, taste, and aroma. Understanding what precisely constitutes this daily elixir is key to appreciating both the art of brewing and the sophisticated science that underpins it.
The Dominant Solvent: Water and the Essence of Extraction
At its most fundamental, a typical cup of black coffee is astonishingly simple in its primary component: over 98.5% water. This seemingly inert liquid, however, is the very medium through which coffee’s intricate character is brought to life. The quality and composition of this water – its mineral content, pH, and temperature – are among the most critical, yet often overlooked, variables in the brewing process. As a highly polar solvent, water selectively extracts compounds from the roasted coffee grounds, dictating the final flavor profile. The journey from dry bean to liquid gold is, at its heart, a sophisticated exercise in selective dissolution.
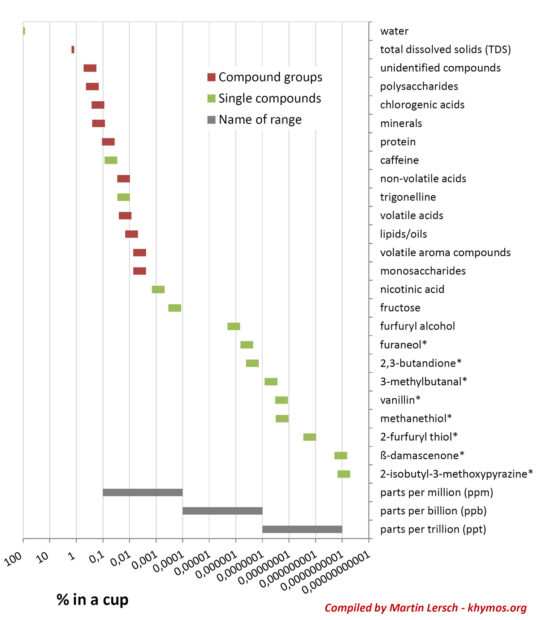
From approximately 60 grams of ground coffee, a mere 12 to 15 grams of solid material are typically extracted into one liter of brewed coffee, representing a Total Dissolved Solids (TDS) concentration of 1.2% to 1.5%. This seemingly small percentage of solids is responsible for the entire sensory experience. The parameters controlling this extraction, such as brew ratio (the proportion of coffee to water), grind size, water temperature, and filter material, are precisely calibrated by skilled baristas and home brewers to optimize the yield of desirable compounds while minimizing the extraction of undesirable ones. These initial physical parameters set the stage for the chemical drama unfolding within the cup.
A Molecular Kaleidoscope: Unraveling Coffee’s Hundreds of Compounds
Delving deeper into these extracted solids reveals a chemical landscape of unparalleled richness. Chromatographic techniques, advanced analytical tools capable of separating individual chemical components, consistently demonstrate that coffee contains hundreds, and potentially thousands, of distinct molecules. While many of these compounds have been identified and characterized, a significant portion remains elusive, posing an ongoing challenge for flavor scientists.

A groundbreaking Aroma Extract Dilution Analysis (AEDA) conducted in 2002, for instance, revealed that a remarkable 13 out of 40 key odorants contributing to coffee’s complex aroma profile were entirely unknown. The difficulty in identifying these compounds often stems from their extremely low concentrations, making it challenging to obtain sufficient sample material for comprehensive structural elucidation. Additionally, some molecules may be unusually large or structurally intricate, further complicating their precise identification. This ongoing mystery highlights the vast unexplored territories within coffee chemistry, suggesting that even after decades of research, much remains to be discovered about what truly defines coffee’s signature scent.
Early attempts to recreate coffee flavor artificially underscored this complexity. A 1996 study managed to synthesize "model" coffees mimicking Arabica and Robusta varieties using just 22 key volatile compounds. While sensory assessors described these synthetic brews as "clearly coffee-like," the phrasing itself implies a significant qualitative gap compared to authentic coffee. This suggests that the full spectrum of coffee aroma requires far more than a couple of dozen compounds, reaffirming the irreplaceable intricacy of nature’s original brew. The pursuit of a truly "perfect" synthetic coffee remains a distant horizon, a testament to the unparalleled complexity of the natural product.
Volatile Compounds: The Architects of Aroma
The sensory experience of coffee is bifurcated into aroma and taste, primarily driven by distinct chemical classes. Volatile compounds, characterized by their ability to readily evaporate and reach the olfactory receptors in the nose, are the architects of coffee’s aroma. These molecules are typically present in minute quantities but possess immense aromatic power. They encompass a diverse range of chemical structures, including pyrazines (nutty, roasted notes), furans (caramel, sweet), aldehydes (fruity, green), ketones (buttery, fruity), thiols (sulfurous, roasted), phenols (smoky, spicy), and esters (fruity). The interplay of these various families creates the vast aromatic palette that distinguishes different roasts, origins, and processing methods.
Non-Volatile Compounds: Sculpting Taste and Body
In contrast, non-volatile compounds primarily influence the taste perceived on the tongue, contributing to sensations such as bitterness, sweetness, acidity, and astringency. Beyond taste, these compounds, along with insoluble particles and emulsified oils, are crucial for coffee’s body and texture – the mouthfeel that adds to its overall richness.
- Acids: Coffee is rich in a variety of acids, notably chlorogenic acids, which are abundant in green coffee and undergo significant transformation during roasting, contributing to both bitterness and astringency. Other important acids include quinic, citric (bright, citrusy notes), malic (green apple), acetic (sharp), and formic acids. The balance and concentration of these acids are critical for the perceived brightness and vibrancy of a coffee.
- Carbohydrates: Sugars like sucrose, glucose, and fructose contribute directly to sweetness, particularly in lighter roasts. Polysaccharides and other soluble oligosaccharides contribute to the body and viscosity of the brew. During roasting, sugars also participate in the Maillard reaction, generating a multitude of flavor and aroma compounds, including those responsible for caramelization and browning notes.
- Lipids (Oils): While sparingly soluble in water, lipids play a crucial role, especially in brewing methods like espresso where they are emulsified to create crema and contribute significantly to body and mouthfeel. These oils carry many of the hydrophobic (non-polar) volatile aroma compounds, acting as a reservoir for flavor.
- Proteins and Amino Acids: These nitrogen-containing compounds are precursors to many flavor molecules through the Maillard reaction and Strecker degradation during roasting. They also contribute to the overall body and potentially some bitter notes.
- Minerals and Salts: Present in trace amounts, minerals such as potassium, magnesium, and calcium influence water hardness and pH, thereby affecting extraction efficiency and the perception of taste.
- Alkaloids: Caffeine: The most famous non-volatile compound, caffeine, is a potent stimulant and a significant contributor to coffee’s characteristic bitterness. Its concentration varies widely depending on the coffee species (Robusta typically has more than Arabica) and preparation method, ranging from tens to hundreds of milligrams per serving.
The Power of Perception: Odor Activity Values (OAVs)
One of the most remarkable aspects of coffee chemistry is the inverse relationship between concentration and sensory impact for certain compounds. While it might seem intuitive that higher concentrations equate to greater influence, this is not always the case in the complex world of olfaction. Our noses possess an extraordinary sensitivity, with each compound having a unique odor threshold – the minimum concentration at which it can be detected. This is where the concept of Odor Activity Value (OAV) becomes critical: the concentration of a compound divided by its odor threshold. A high OAV indicates a strong contribution to the overall aroma, even if the compound is present in minuscule amounts.
Beta-Damascenone: A Potent Aroma Molecule
A prime example of a compound with an exceptionally high OAV is beta-damascenone. This potent odorant can be present in coffee at concentrations as low as one part per trillion (ppt), yet it exerts a significant influence on the overall flavor profile. In fact, studies have identified beta-damascenone as one of the top five most impactful odorants in coffee, contributing complex fruity, floral, rose, and blackcurrant notes. Its presence underscores the sophisticated interplay of compounds at the molecular level that creates coffee’s rich sensory experience.
Beta-damascenone belongs to a family of molecules known as rose ketones, first identified in rose oil in the 1960s. These compounds revolutionized the fragrance industry, famously playing a "first violin" role in Dior’s iconic perfume, Poison. The name "damascenone" itself is derived from the Damask rose (Rosa × damascena), highlighting its origins in natural floral essences. The Good Scents Company, a comprehensive database for flavors and fragrances, lists an impressive array of sensory descriptors for beta-damascenone, including woody, spicy, fruity, sweet, apple, plum, and tobacco notes, demonstrating its versatility and complexity. The discovery of its significant role in coffee further illustrates the fascinating cross-pollination of flavor and fragrance chemistry across seemingly disparate natural products. The ability to accurately measure odor thresholds and quantify compounds at such ultralow concentrations is a testament to the advancements in analytical chemistry, yet it remains a challenging and labor-intensive endeavor, suggesting that more key odorants in coffee await discovery.
Extraction Dynamics: Polar vs. Non-Polar Solutes
The science of coffee extraction is fundamentally governed by the polarity of its constituent compounds. Coffee’s vast chemical inventory can be broadly categorized into polar and non-polar compounds, each with distinct solubility characteristics in water, the primary solvent.
- Polar Compounds: This group includes acids, minerals, proteins, and carbohydrates (sugars and soluble oligosaccharides). These compounds share a common