Every morning ritual involving a cup of coffee is, in essence, a sophisticated interaction with a complex chemical concoction, far surpassing the simple perception of a dark, stimulating beverage. While appearing deceptively simple, black coffee is over 98.5% water, yet the remaining 1.2-1.5% of total dissolved solids (TDS) represents a staggering array of hundreds, if not thousands, of unique molecules. These extracted compounds, typically amounting to 12-15 grams per liter of brewed coffee derived from approximately 60 grams of ground beans, are the true architects of coffee’s celebrated color, taste, and aroma, making it one of the most chemically intricate consumables known to humankind.
A Deep Dive into Coffee’s Molecular Complexity
The scientific pursuit to unravel coffee’s chemical identity has been a journey marked by both remarkable discoveries and persistent enigmas. Utilizing advanced chromatographic techniques, researchers can separate individual chemical components, revealing a vast molecular landscape. This complexity is so profound that, as far back as a 2002 aroma extract dilution analysis (AEDA), it was found that 13 of the 40 identified key odorants in coffee remained chemically unknown. The difficulty in fully characterizing these molecules often stems from their presence in extremely low concentrations, making isolation for structural elucidation challenging, or from their large molecular size, which complicates the analytical process.
Early attempts to replicate coffee’s essence underscore this complexity. A 1996 study, for instance, managed to create "model" coffees mimicking Arabica and Robusta varieties using just 22 key volatile compounds. While assessors described these synthetic versions as "clearly coffee-like," the consensus among flavor scientists is that such a limited palette falls far short of perfectly capturing the nuanced, multi-dimensional aroma profile of a naturally brewed cup. This ongoing challenge highlights the immense gap between a basic "coffee-like" impression and the rich, authentic experience delivered by natural extraction, reinforcing the notion that perfect synthetic competition remains a distant prospect.
The Bifurcation of Flavor: Volatiles and Non-Volatiles
The myriad compounds in coffee can be broadly categorized into volatile and non-volatile components, each playing a distinct yet interconnected role in the overall sensory experience. Volatile compounds, characterized by their ability to readily evaporate, are primarily responsible for the aroma detected by our olfactory receptors. These are the molecules that waft from a freshly brewed cup, creating anticipation and contributing to the "nose" of the coffee. In contrast, non-volatile compounds interact with taste receptors on the tongue, contributing to fundamental tastes such as bitterness, sweetness, acidity, and astringency. Beyond taste, non-volatile components, including various oils and insoluble microscopic particles, are crucial for defining the coffee’s body and texture, impacting mouthfeel and viscosity.
Within the soluble components of a typical cup, scientists have identified a diverse range of chemical groups. These include various carbohydrates (sugars and soluble oligosaccharides), a spectrum of acids (e.g., chlorogenic acids, quinic acids), essential minerals and salts, proteins, trace lipids, and, of course, the stimulant caffeine. Furthermore, a substantial portion remains "unidentified," often comprising larger, complex molecules like melanoidins, which are products of the Maillard reaction during roasting and contribute significantly to coffee’s characteristic color and perceived bitterness. The intricate interplay of these identified and unidentified components creates the unique sensory fingerprint of each coffee.

The Unseen Architects of Aroma: Concentration vs. Impact
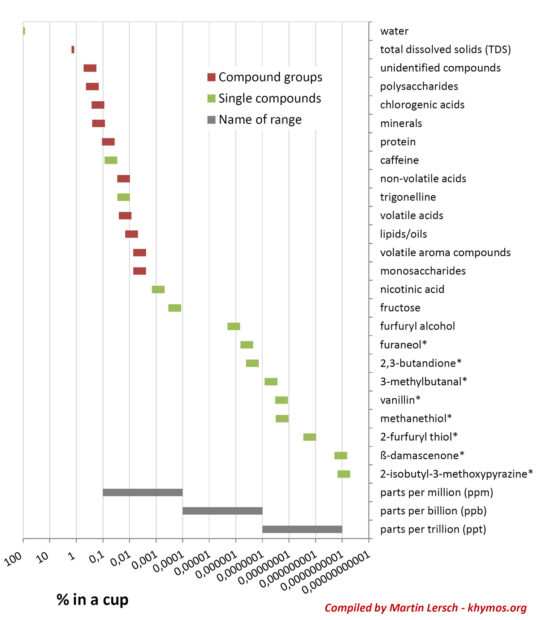
Perhaps one of the most fascinating aspects of coffee chemistry is the disproportional relationship between a compound’s concentration and its sensory impact. A visual representation of coffee’s composition, often plotted on a logarithmic scale, reveals a concentration range spanning more than nine orders of magnitude. This vast spectrum means that while some compounds, like water, exist at over 98%, others are present in parts per trillion (ppt). Counter-intuitively, a lower concentration does not necessarily equate to a lower impact. This phenomenon is explained by the concept of odor activity value (OAV), which considers a compound’s concentration relative to its odor threshold – the minimum concentration at which it can be detected by the human nose.
This principle means that a compound present at an astonishingly low concentration, perhaps 0.0000000001%, can be as vital to the overall flavor profile as another compound present at 0.01%. Our olfactory system possesses an extraordinary sensitivity to certain molecules, giving them disproportionate influence. A prime example is β-damascenone, a potent odorant found in coffee at concentrations as low as 1 part per trillion (ppt). Despite its minute presence, β-damascenone is consistently ranked among the top five most impactful odorants in coffee. Its flavor profile is described as sweet, fruity, floral, woody, tobacco, and tea-like, contributing significantly to coffee’s complex bouquet.
The Story of β-Damascenone and Rose Ketones
The discovery and recognition of β-damascenone’s importance in coffee flavor highlight a broader scientific narrative. β-damascenone belongs to a class of compounds known as rose ketones, first identified in rose oil in the 1960s. These molecules are renowned for their intense and characteristic floral notes. The name "damascenone" itself is derived from the Damask rose (Rosa × damascena), a flower historically cultivated for its prized fragrance.
The commercial impact of rose ketones was profoundly demonstrated by the flavor and fragrance company Firmenich, which pioneered their use. β-damascenone, in particular, became a foundational component in iconic perfumes, most notably Dior’s "Poison," where it played a central role alongside other rose ketones, shaping an entire era of perfumery. The journey of β-damascenone from a key component in luxury fragrances to a recognized essential contributor to coffee’s complex aroma underscores the interdisciplinary nature of flavor science. The precision required to measure odor thresholds and quantify such trace compounds is immense, suggesting that ongoing advancements in analytical chemistry may yet uncover additional, equally potent, and previously unknown key odorants in coffee.
Extraction Dynamics: Polar vs. Non-Polar Compounds
Understanding coffee’s chemical makeup also necessitates distinguishing between polar and non-polar compounds, as their differing solubilities profoundly influence extraction dynamics during brewing. Polar compounds, which include acids, minerals, proteins, and carbohydrates, are highly water-soluble. Water, being a polar solvent, is exceptionally efficient at extracting these compounds, typically yielding more than 75% of them into the beverage. These components largely contribute to coffee’s acidity, sweetness, and mineral balance.
Conversely, non-polar compounds, encompassing many volatile aroma molecules and lipids (oils), exhibit low solubility in water. Consequently, when brewing coffee with water, only a limited fraction—typically 10-30%—of these non-polar compounds are extracted. This differential extraction efficiency has significant implications for brewing methods and the final flavor profile. For instance, cold brewing, which uses lower temperatures, tends to extract fewer acidic (polar) compounds, resulting in a smoother, less bitter cup. The potential for exploring alternative, less polar solvents, such as ethanol, for specialized coffee extractions opens avenues for unlocking an even broader spectrum of coffee’s inherent flavors and aromas, a concept already explored in the production of coffee liqueurs and flavorings.
The Limitations of Total Dissolved Solids (TDS)
While Total Dissolved Solids (TDS) measurements are widely used within the coffee industry as a practical indicator of brew strength, providing a quantitative measure of how much "coffee stuff" is in the water, it offers a surprisingly incomplete picture of flavor. The typical TDS range of 1.2-1.5% in black coffee is influenced primarily by the most abundant soluble compounds. However, the concentration ranges of these compounds highlight that only a few major groups significantly impact the overall TDS reading.
The fundamental flaw in relying solely on TDS as a proxy for flavor quality lies in the non-uniform extraction rates of different compound classes. As discussed, polar and non-polar compounds extract at vastly different efficiencies. Furthermore, many of the most impactful aroma compounds, like β-damascenone, exist at such minute concentrations that their contribution to the overall TDS is negligible, despite their profound sensory influence. Therefore, while a TDS measurement can guide consistency in strength, it is a very poor predictor of the nuanced aroma and complex flavor profile of a cup of coffee. This reinforces the enduring truth that the most sophisticated analytical instrument for evaluating coffee quality remains the human sensory system – our nose and palate – whose unparalleled sensitivity and integrative capacity continue to be the gold standard for exploring the wonders of coffee extraction.
Implications and Future Directions in Coffee Science
The ongoing scientific dissection of coffee’s chemical composition holds profound implications for various sectors, from specialty coffee producers to global flavor manufacturers. For the specialty coffee industry, this deeper understanding informs precise roasting profiles, optimized brewing parameters, and innovative processing techniques designed to accentuate specific desirable flavor notes. Roasters can manipulate heat and time to encourage or suppress the formation of certain volatile compounds, while baristas can fine-tune grind size, water temperature, and contact time to selectively extract desired soluble components.
In food science, coffee serves as a quintessential model for studying complex flavor systems, pushing the boundaries of analytical chemistry and sensory evaluation. Research into coffee’s components continues to inspire advancements in other areas of food and beverage development. The quest to fully characterize the "unknown" compounds represents a frontier for academic research, potentially leading to the discovery of novel flavor molecules with applications far beyond coffee itself.
For consumers, an appreciation of this chemical complexity elevates the experience of drinking coffee from a mere habit to a connoisseurship. Understanding that a simple brew is a symphony of hundreds of molecules, each playing its part, fosters a greater appreciation for the artistry of coffee cultivation, processing, and preparation. While the dream of "synthetic coffee" might exist in some corners, the sheer, unparalleled complexity and synergistic interaction of natural compounds ensure that the authentic, brewed cup, with its blend of known and mysterious molecules, will continue to reign supreme, offering an inexhaustible source of sensory delight and scientific inquiry. As analytical techniques become even more sophisticated, the future promises an even clearer picture of what truly resides in that daily cup, allowing for ever more refined and exquisite coffee experiences.