The creation of ginger milk curd, known in Chinese as 姜汁撞奶 (Jiāng zhī zhuàng nǎi), stands as a remarkable culinary phenomenon, transforming three simple ingredients—milk, ginger, and sugar—into a delicate, tender gel within minutes. This traditional dessert, cherished for its smooth texture and pungent ginger kick, has long baffled home cooks and even seasoned chefs due to its notoriously inconsistent results. What appears to be a straightforward preparation often devolves into a frustrating series of failures, giving rise to numerous kitchen myths and contradictory instructions that obscure the underlying scientific principles.
A Culinary Heritage: The Origins and Appeal of Ginger Milk Curd
Ginger milk curd boasts a rich history, believed to have originated in the Shawan region of Guangzhou, China, during the Ming Dynasty. Legend has it that an elderly woman, suffering from a stomach ailment, found relief after consuming milk curdled with ginger juice. The recipe quickly gained popularity, evolving into a beloved dessert across Cantonese-speaking regions and beyond. Its appeal lies not only in its unique flavor profile—the warmth of ginger complementing the creamy sweetness of milk—but also in its perceived health benefits, as ginger is traditionally revered for its digestive and anti-inflammatory properties.
Despite its cultural prominence, the preparation of ginger milk curd has remained largely an art rather than a science for generations of home cooks. The process hinges on precise conditions that are often not explicitly understood, leading to a high rate of failure. Reports from home kitchens frequently recount experiences ranging from success rates as low as 50% to achieving the desired gel only after numerous attempts, or even abandoning the traditional method altogether in favor of stabilizers like egg whites. These anecdotal accounts underscore the pervasive sense of mystery surrounding this seemingly simple dish, reinforcing the notion that its success relies on a blend of skill and an elusive "pinch of luck."
The Labyrinth of Kitchen Myths: Contradictory Advice Abounds

The widespread difficulty in consistently producing ginger milk curd has fostered a fertile ground for "kitchen myths." These myths often emerge when cooks attribute success or failure to random variables in their process, rather than identifying the true scientific factors at play. As researchers delve into the mechanics of gel formation in foods without external hydrocolloids, the case of ginger milk curd frequently emerges as a prime example of culinary complexity masked by apparent simplicity.
A review of traditional and online recipes reveals a bewildering spectrum of advice, often specific yet contradictory. For instance, some recipes advocate for:
- Milk Temperature: "Heat milk to boiling, then cool slightly" versus "Heat milk to exactly 70°C, no more, no less."
- Ginger Juice Preparation: "Use only the very first squeeze of juice" versus "Any ginger juice is fine, just make sure it’s fresh."
- Mixing Technique: "Stir vigorously immediately after pouring" versus "Do NOT stir at all, let it set undisturbed."
- Setting Conditions: "Place in the fridge immediately" versus "Allow to set at room temperature for 10-15 minutes."
- Ingredient Ratios: "More ginger for a firmer set" versus "Too much ginger will prevent setting."
Such conflicting instructions highlight the deep-seated lack of understanding regarding the precise conditions required for the enzymatic reaction. Without a clear scientific framework, home cooks resort to trial and error, inadvertently creating and perpetuating these myths based on correlation rather than causation. This phenomenon is a classic illustration of cognitive bias in culinary contexts, where an outcome is linked to the most salient change, regardless of its actual relevance.
Unveiling the Scientific Mechanism: Ginger Proteases and Casein Coagulation
The secret to ginger milk curd lies in a sophisticated biochemical interaction, placing it firmly within the category of enzyme-curdled dairy products, much like cheese. Historically, cheese making has relied on rennet, an enzyme complex found in the stomachs of young mammals, with chymosin (also known as rennin) being its primary active component. Chymosin is a proteolytic enzyme, or protease, meaning it is capable of breaking down proteins into smaller fragments.
Ginger, it turns out, contains its own potent proteolytic enzymes, collectively known as ginger proteases (GP), with zingipain (EC 3.4.22.67) being a key player. These enzymes are the tireless workers responsible for initiating the gelling process. However, unlike the broad activity range of some proteases, ginger proteases are exceptionally sensitive to temperature, which is the crux of the ginger milk curd mystery.

The Critical Temperature Window: Precision is Paramount
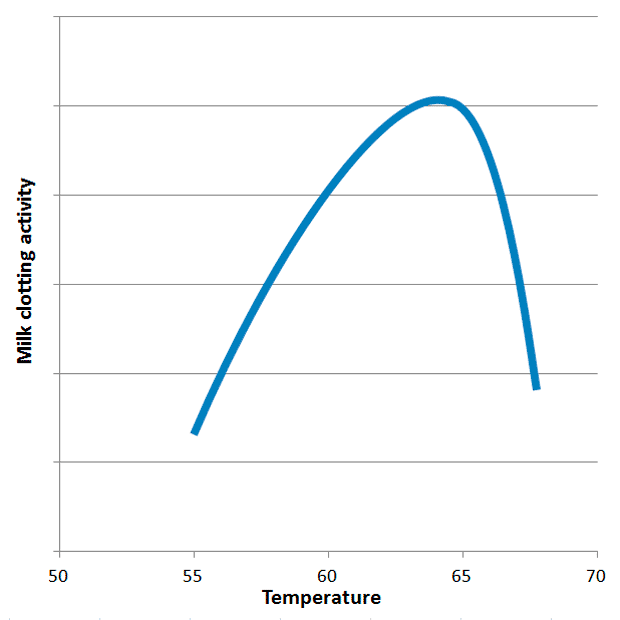
Research into ginger proteases reveals a narrow, critical temperature window for optimal milk clotting activity (MCA). The activity of GP peaks sharply around 63°C, declining rapidly both above 65°C and below 60°C. Above 70°C, the enzymes quickly denature, meaning they are irreversibly destroyed and lose their ability to curdle milk. This precise temperature requirement is the primary reason for the high failure rate among home cooks, many of whom unknowingly overheat or underheat their milk. While GP may exhibit general proteolytic activity outside this optimal range, its specific milk-clotting action, which targets a particular milk protein, is highly temperature-dependent.
This specificity also has broader implications. Scientists have explored various plant extracts for their milk-clotting properties as potential alternatives to animal rennet, particularly for vegetarian cheese production. Ginger, along with kiwi and melon, has been identified as containing proteases with a relatively high MCA/PA (milk clotting activity to general proteolytic activity) ratio, making them promising candidates, though typically not as efficient as chymosin. Interestingly, kiwi proteases have an optimal MCA at 40°C, while melon proteases peak at 70°C, showcasing the diverse temperature sensitivities of plant-derived enzymes.
Casein: The Building Blocks of the Gel
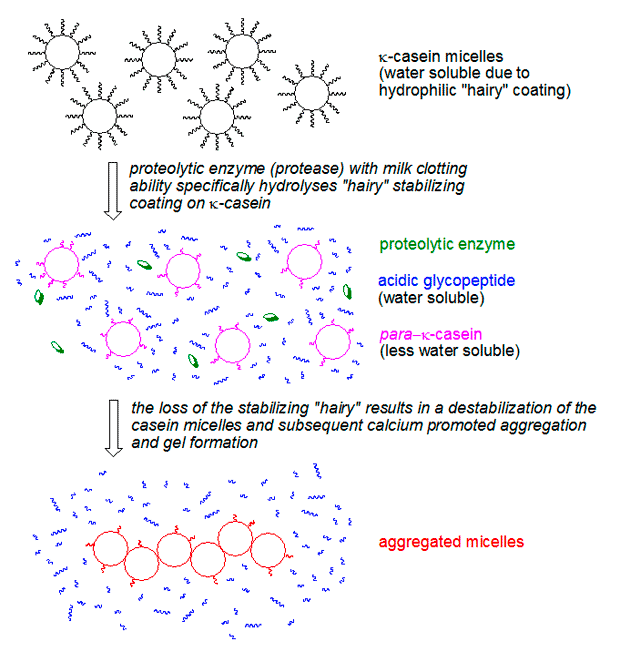
While ginger proteases act as the catalysts, the actual structural components of the gel are casein proteins, which constitute about 80% of the protein in milk. Casein proteins exist in milk as large, spherical structures called micelles, held together by calcium phosphate bridges. The stability of these micelles, preventing them from aggregating and precipitating, is largely due to a specific protein called κ-casein (kappa-casein).
κ-casein is unique because it has a hydrophilic (water-loving) region that extends outwards from the micelle surface, forming a "hairy" layer. This layer creates steric hindrance and electrostatic repulsion, effectively keeping the micelles dispersed and stable in the aqueous milk environment. Without this protective layer, the casein micelles would naturally aggregate.

This is precisely where ginger proteases (and chymosin in rennet) come into play. These enzymes specifically cleave the hydrophilic "hairy" portion of the κ-casein, leaving behind a less soluble part called para-κ-casein. Once "shaved" of their protective outer layer, the now hydrophobic casein micelles lose their repulsive forces. The abundant calcium ions naturally present in milk then act as bridges, facilitating the aggregation and coagulation of these denuded micelles. This rapid aggregation forms the three-dimensional protein network that constitutes the tender, fragile gel within minutes. The resulting gel is known for its tendency to undergo syneresis, where liquid (whey) is expelled from the shrinking gel matrix, a common characteristic of enzyme-coagulated curds.
Optimizing Gel Strength: Beyond Temperature
Further research indicates that other factors significantly influence the strength and quality of the ginger milk curd. For instance, heating milk to temperatures above 65°C, even if subsequently cooled to the optimal range, can negatively impact gel formation. This is because excessive heat can cause other milk proteins, particularly whey proteins like lactoglobulins, to denature and precipitate onto the κ-casein micelles. This undesirable coating interferes with the specific cleavage action of ginger proteases and the subsequent aggregation process, leading to a weaker or non-existent gel.
Similarly, milk fat can also impede gel formation. Skimmed milk, with its lower fat content, generally yields a stronger and more consistent gel compared to whole milk. This is likely due to fat globules physically interfering with the casein micelle network formation. Moreover, since calcium plays a crucial role in bridging the "shaved" micelles, a higher concentration of calcium can lead to a firmer gel, though excessive calcium could also lead to a grainy texture.
The Volatile Nature of Ginger Juice: A Race Against Time
Another critical factor often overlooked in traditional recipes is the stability of the ginger juice itself. Freshly squeezed ginger juice is a powerhouse of enzymes, but its efficacy is fleeting. Ginger proteases have a remarkably short half-life—approximately 20 minutes at 30°C. This means that in a warm kitchen, half of the enzyme activity can be lost within 20 minutes of grating and squeezing the ginger. After another 20 minutes, only 25% of the original activity remains. This rapid degradation explains why preparing ginger juice in advance or storing it at room temperature often leads to failed curd.
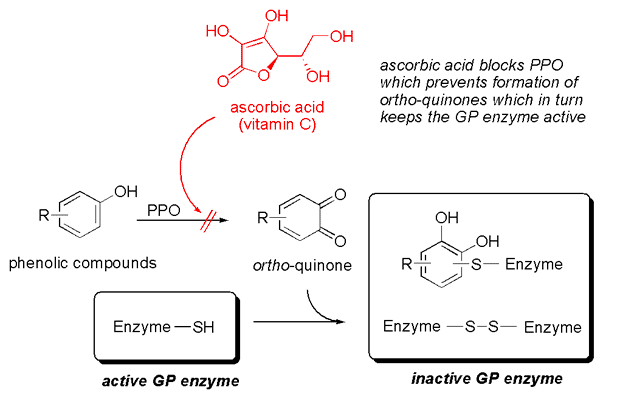
The culprit behind this instability is another enzyme present in ginger: polyphenol oxidase (PPO). PPO is notorious for causing enzymatic browning in fruits and vegetables, like apples. When ginger is grated, cell walls are broken, releasing PPO which then oxidizes phenolic compounds into ortho-quinones. These quinones are highly reactive and can bind to and inactivate the delicate ginger protease enzymes, effectively sabotaging their milk-clotting potential.
Fortunately, there’s a simple scientific solution: ascorbic acid, commonly known as Vitamin C. Ascorbic acid acts as an antioxidant, blocking the activity of PPO. By adding a small pinch (approximately 0.2% by weight) of ascorbic acid to freshly prepared ginger juice, the PPO activity is inhibited, thereby preserving the integrity and activity of the ginger proteases. This trick allows for the ginger juice to be prepared slightly in advance without significant loss of enzymatic power, adding another layer of "foolproof" reliability to the recipe.
A Foolproof Recipe: Bridging Science and Culinary Practice
Armed with this scientific understanding, a consistently successful ginger milk curd is no longer a matter of luck but precise execution. The most indispensable tool in this endeavor is a digital kitchen thermometer, ensuring the milk reaches the critical temperature window.
Foolproof Ginger Milk Curd Recipe
-
Ingredients:
- 250 mL skimmed milk
- 18 g fresh ginger juice (approximately 31 g peeled ginger or 43 g raw ginger)
- 20 g sugar
-
Instructions:
- Prepare the Milk: Combine skimmed milk and sugar in a saucepan. Heat carefully, monitoring with a digital thermometer, until the mixture reaches precisely 65°C. Remove immediately from heat.
- Prepare the Ginger Juice: While the milk is heating, peel fresh ginger and microplane it to maximize juice extraction. Squeeze out the juice using a cheesecloth or a garlic press. Measure 18 g of juice accurately. (For optimal enzyme stability if preparing slightly in advance, add a tiny pinch of ascorbic acid powder, about 0.05g, to the ginger juice).
- Combine and Set: Pour the measured ginger juice into the bottom of a serving bowl. Immediately and carefully, pour the 65°C milk from a moderate height (about 15-20 cm) into the bowl containing the ginger juice. This height ensures sufficient initial mixing without the need for stirring.
- Do NOT Stir: Crucially, do not stir the mixture after pouring. Stirring can disrupt the delicate formation of the casein network, leading to a weaker or broken gel.
- Allow to Set: Leave the bowl undisturbed at room temperature. Within 5-10 minutes, a tender, fragile gel will form, firm enough to support the weight of a spoon.
- Serve: The curd can be served immediately, warm, or chilled in the refrigerator for a refreshing cold dessert.
This recipe utilizes a milk-to-ginger juice ratio of approximately 14:1. While more ginger juice can be used for a stronger ginger flavor, excessive amounts might overwhelm the palate or, if the protease concentration becomes too high relative to available casein, could paradoxically lead to a weaker or grainy curd. Conversely, reducing the ginger juice below a certain threshold would result in insufficient enzyme activity for proper gel formation.
Broader Implications: From Kitchen to Industry
The scientific demystification of ginger milk curd holds significant implications beyond the home kitchen. For culinary professionals, this knowledge translates into consistent product quality and the ability to innovate with traditional recipes. Understanding the precise temperature, enzyme activity, and protein interactions allows for greater control over texture and flavor, opening avenues for modern interpretations of classic dishes.
In the food industry, this research contributes to the growing interest in plant-based coagulants. As demand for vegetarian and vegan products rises, identifying and characterizing plant proteases with milk-clotting capabilities becomes increasingly important. Ginger proteases, along with those from kiwi and melon, offer viable alternatives to traditional animal rennet, potentially enabling the production of new types of plant-based cheeses and dairy alternatives. The stability issues of ginger proteases, as elucidated by the PPO-ascorbic acid interaction, also provide valuable insights for industrial processing, storage, and formulation of enzyme-containing ingredients.
Future Directions and Culinary Experimentation
While a robust understanding of ginger milk curd has been achieved, several avenues remain open for further exploration, inviting both scientific inquiry and home-based experimentation:
- Alternative Milks: Could ginger milk curd be successfully made with plant-based milks (e.g., soy milk, almond milk, oat milk) which have different protein compositions and pH levels? This would require investigating the interaction of ginger proteases with alternative protein sources.
- Ginger Variety and Maturity: Does the variety or maturity of ginger significantly impact its protease activity? Different ginger types might yield varying enzyme concentrations and activity levels.
- Optimal pH: What is the precise optimal pH for ginger protease activity in milk? While temperature is key, pH can also strongly influence enzyme kinetics.
- Calcium Fortification: How does adding exogenous calcium salts (e.g., calcium chloride) affect the gel strength and texture, particularly in milks with naturally lower calcium content?
- Fat Content Revisited: A more detailed study on the precise impact of varying fat percentages in milk on gel formation and stability could offer nuanced insights.
- Ascorbic Acid Concentration: What is the minimal effective concentration of ascorbic acid needed to stabilize ginger proteases without affecting flavor or texture?
The journey from a mysterious culinary tradition to a scientifically understood process underscores the intricate interplay of chemistry and cooking. By bridging the gap between kitchen folklore and empirical evidence, ginger milk curd transforms from an unpredictable delight into a triumph of precise culinary science, offering consistent enjoyment and opening new possibilities for food innovation.









