A seemingly simple cup of black coffee, often perceived as a mere blend of water and ground beans, is in fact an extraordinarily complex chemical solution, a symphony of hundreds, if not thousands, of distinct molecules contributing to its iconic color, taste, and aroma. More than 98.5% water, the remaining 1.2% to 1.5% consists of total dissolved solids (TDS), meaning a single liter of brewed coffee contains approximately 12 to 15 grams of compounds extracted from roughly 60 grams of coffee grounds. This intricate chemical profile is the subject of ongoing scientific inquiry, revealing that even with advanced analytical techniques, a significant portion of coffee’s sensory magic remains a tantalizing mystery.
The Evolving Science of Coffee Analysis
The journey to understand coffee’s chemical composition mirrors the evolution of food science itself. For centuries, coffee consumption was driven by cultural tradition and empirical observation, with roasters and brewers perfecting their craft through trial and error. However, the advent of sophisticated analytical chemistry in the 20th century, particularly chromatographic techniques such as Gas Chromatography-Mass Spectrometry (GC-MS) and High-Performance Liquid Chromatography (HPLC), revolutionized our ability to dissect complex mixtures. These tools allowed scientists to separate individual chemical components, revealing the sheer scale of molecular diversity in coffee. Early studies in the mid-to-late 20th century began to identify major constituents like caffeine, chlorogenic acids, and various sugars. The 1990s and early 2000s saw a significant acceleration in the characterization of volatile aroma compounds, which are crucial for coffee’s distinctive fragrance. Researchers like those cited in the original article, including Grosch and Schieberle, have been at the forefront of this detailed chemical mapping, pushing the boundaries of what is known about this globally cherished beverage.
Deconstructing the Brew: Major Soluble Solids
The 12-15 grams of solids per liter of brewed coffee comprise a diverse array of chemical classes, each playing a role in the beverage’s overall profile. These can be broadly categorized as carbohydrates, acids, minerals/salts, proteins, lipids, caffeine, and a vast collection of volatile aroma compounds.
Carbohydrates, including various sugars (like sucrose, fructose, and glucose) and soluble oligosaccharides, contribute to coffee’s sweetness and body. Their presence is significantly influenced by the roasting process, as sugars undergo caramelization and Maillard reactions, forming new compounds that add to flavor and color.
Acids are paramount to coffee’s vibrancy and complexity. Chlorogenic acids (CGAs) are particularly abundant in green coffee beans and are partially hydrolyzed during roasting into quinic and caffeic acids, contributing to bitterness and astringency. Other organic acids like citric, malic, acetic, and lactic acids impart varying degrees of sourness and fruity notes, creating a balanced and often bright taste profile. The type and concentration of these acids are heavily dependent on the coffee varietal, origin, and processing methods.
Minerals and salts, extracted from the beans, contribute to the overall mouthfeel and can subtly influence flavor perception. Elements like potassium, magnesium, and calcium are typically present. Proteins and peptides also contribute to body and crema formation, particularly in espresso, though their concentration in brewed coffee is generally lower than other components. Lipids, or oils, contribute significantly to coffee’s body, mouthfeel, and the stability of crema. While largely non-polar and less soluble in water, a portion of these oils is extracted, especially in brewing methods that do not use paper filters.

Caffeine, perhaps the most famous compound in coffee, is a purine alkaloid known for its stimulant properties. It also contributes a distinct bitterness to the taste profile, although its sensory impact is often balanced by other compounds. The concentration of caffeine varies widely depending on the coffee species (Arabica typically has less than Robusta), roast level, and brewing method.
The Symphony of Aroma: Volatile Compounds
The true magic of coffee, its captivating aroma, resides in its volatile compounds – molecules that readily evaporate and are detected by our olfactory system. Scientists have identified hundreds, if not thousands, of these compounds, yet the full picture remains elusive. An Aroma Extract Dilution Analysis (AEDA) conducted in 2002, for instance, dramatically highlighted this complexity by revealing that 13 of the 40 key odorants in coffee were, at that time, entirely unknown to science [1]. This points to the formidable challenge of complete chemical characterization. These unidentified compounds are often present in exceedingly low concentrations, making isolation and structural elucidation difficult, or they might be large, complex molecules whose structures are inherently challenging to resolve.
The sheer number of volatile compounds and their synergistic interactions create coffee’s unique aromatic fingerprint. A study from 1996 illustrated this complexity when researchers attempted to create "model" coffees mimicking Arabica and Robusta using 22 key volatile compounds [2]. While tasters described these synthetic brews as "clearly coffee-like," they were far from perfectly replicating the nuanced, rich aroma of natural coffee. This suggests that the subtle interplay of numerous compounds, many at trace levels, is essential for a truly authentic coffee experience. Common aroma families include pyrazines (responsible for roasted, nutty notes), furans (caramel, sweet), aldehydes (fruity, green), ketones (buttery, sweet), and sulfur-containing compounds (roasted, sometimes savory). Each contributes a unique note to the overall olfactory tapestry.
The Dance on the Tongue: Non-Volatile Compounds and Taste
While volatile compounds stimulate our sense of smell, non-volatile compounds interact with taste receptors on the tongue, contributing to the fundamental tastes of bitterness, sweetness, acidity, and astringency. Many of these non-volatiles also contribute to the coffee’s body and texture.
Bitterness in coffee is primarily attributed to caffeine, chlorogenic acid lactones, and melanoidins. Melanoidins are large, complex polymers formed during the Maillard reaction (non-enzymatic browning) during roasting. They also contribute significantly to coffee’s dark color and body, and represent a substantial portion of the "unidentified compounds" in the cup.
Sweetness comes from residual sugars and the perception of certain volatile compounds. Acidity, as mentioned, is shaped by organic acids like citric, malic, and acetic acids, which provide brightness and complexity. Astringency, often described as a drying sensation in the mouth, is primarily due to phenolic compounds, including tannins and specific chlorogenic acid derivatives. The balance of these tastes is crucial for a well-rounded and enjoyable cup of coffee, often requiring careful control during sourcing, roasting, and brewing.
The Potent Players: Trace Compounds and Odor Activity Value (OAV)
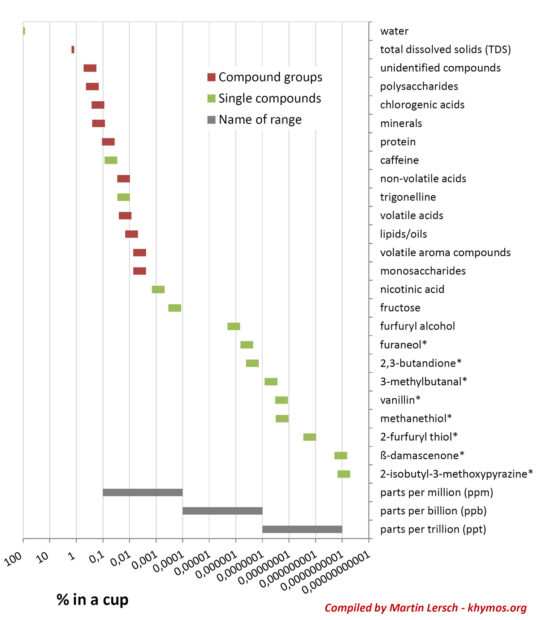
One of the most profound insights into coffee’s flavor lies in understanding the concept of Odor Activity Value (OAV). OAV is calculated by dividing the concentration of a compound by its odor threshold – the minimum concentration at which it can be detected by the human nose. This metric is critical because it explains why compounds present in minute quantities can have a disproportionately large impact on overall flavor. As the original article highlights, a compound present at 0.0000000001% (parts per trillion) can be as significant as one present at 0.01% if our nose is exceptionally sensitive to it. The logarithmic scale used in scientific diagrams illustrates this vast range, spanning more than nine orders of magnitude in concentration.
A prime example of such a potent trace compound is beta-damascenone. This remarkable molecule, present in coffee at concentrations as low as 1 part per trillion (ppt), is recognized as one of the top five most impactful odorants [1]. Its sensory profile is complex and alluring, often described as fruity (apple, plum, blackcurrant), floral (rose), tea-like, and honeyed. Beta-damascenone belongs to a group known as rose ketones, first identified in rose oil in the 1960s. Its discovery was a landmark in the flavor and fragrance industry, with companies like Firmenich pioneering its use. Famously, beta-damascenone and other rose ketones became cornerstone ingredients in iconic perfumes, such as Dior’s "Poison," demonstrating their extraordinary power to evoke rich and complex scents. Its presence in coffee, therefore, adds a layer of sophisticated fruitiness and floral notes that are crucial to the overall aromatic character, despite its minuscule concentration. The ability of the human olfactory system to detect such compounds at such low levels is a testament to its incredible sensitivity and complexity. Other highly potent odorants in coffee include 2-furfurylthiol (roasted, coffee-like), guaiacol (phenolic, smoky), and various pyrazines.
The Challenge of Extraction: Polar vs. Non-Polar Compounds
The brewing process itself is a selective extraction, dictated by the polarity of the solvent (water) and the compounds in the coffee grounds. Water is a highly polar solvent, meaning it efficiently dissolves other polar compounds. Consequently, over 75% of the polar compounds in coffee beans – including acids, minerals, carbohydrates (sugars and soluble oligosaccharides), and caffeine – are readily extracted into the brew [2]. This explains why these components form the backbone of coffee’s taste and strength.
However, water is far less effective at extracting non-polar compounds, such as lipids (oils) and many volatile aroma molecules. Typically, only 10% to 30% of these non-polar compounds make their way into the cup during water-based brewing. This differential extraction yield has significant implications for how different brewing methods produce varying flavor profiles. For instance, espresso, brewed under high pressure, extracts more oils and finer particulates, contributing to its distinct body and crema. Brewing with a paper filter, conversely, can trap many non-polar oils, resulting in a cleaner cup with a brighter acidity. This understanding of polarity also opens doors for alternative extraction methods; for example, using less polar solvents like ethanol could yield entirely different chemical profiles, though such methods are generally reserved for specialized applications like flavor extracts or decaffeination rather than everyday brewing.
Beyond TDS: The Future of Coffee Analysis and Craft
While Total Dissolved Solids (TDS) measurements provide a convenient metric for coffee strength, they offer a very limited view into the beverage’s nuanced flavor and aroma. As the detailed analysis of coffee’s composition reveals, TDS predominantly reflects the concentration of major polar compounds. It fails to account for the differential extraction rates of polar versus non-polar compounds and, crucially, provides no insight into the presence or impact of highly potent trace odorants like beta-damascenone. A TDS reading cannot predict the delicate balance of sweetness, acidity, bitterness, and the myriad of aromatic notes that define a truly exceptional cup of coffee.
The ongoing research into coffee chemistry underscores a critical interplay between scientific rigor and sensory artistry. Leading food chemists continue to push the boundaries, employing increasingly sensitive analytical tools to identify more of the "unknown" odorants and understand their complex interactions. This scientific endeavor provides valuable insights for the entire coffee industry, from producers optimizing cultivation and processing, to roasters perfecting their craft, and baristas refining brewing parameters. For instance, a deeper understanding of specific aroma precursors could lead to new roasting techniques that enhance desired flavor notes. Similarly, knowledge of extraction dynamics can inform the design of more efficient and flavor-optimized brewing equipment.
Ultimately, despite the sophistication of modern chemistry, the human nose and palate remain the most superb and indispensable analytical detectors for coffee. The appreciation of coffee’s wonders is an experience that transcends mere chemical data, inviting us to continually explore the rich, complex, and ever-evolving sensory landscape contained within each cup. As research progresses, new discoveries are bound to emerge, further enriching our understanding and appreciation of this universally beloved beverage, ensuring that the exploration of coffee’s chemical tapestry remains an exciting frontier.









