The ubiquitous cup of black coffee, often perceived as a simple beverage, is in fact a complex aqueous solution, a meticulously crafted symphony of hundreds, if not thousands, of chemical compounds. Far from being merely a dark, invigorating liquid, each brew represents a sophisticated extraction of organic and inorganic molecules from roasted coffee beans, a process that culminates in the distinctive aroma, taste, and mouthfeel cherished by billions worldwide. This intricate chemical dance, largely hidden from the naked eye, is the subject of intense scientific scrutiny, as researchers endeavor to decode the precise molecular architecture that defines one of the world’s most popular drinks.
The Foundations of Flavor: Water and Total Dissolved Solids
At its most fundamental, black coffee is overwhelmingly composed of water, typically exceeding 98.5% of the beverage’s volume. This significant proportion underscores water’s role not merely as a solvent but as the primary medium dictating the extraction process. The remaining 1.2% to 1.5% comprises what are known as Total Dissolved Solids (TDS) – a critical metric in the coffee industry for gauging brew strength. To put this into perspective, a single liter of brewed coffee contains between 12 to 15 grams of these extracted solids, derived from approximately 60 grams of ground coffee.
The selection and composition of brewing water are paramount. Water rich in certain minerals, such as magnesium and calcium, can enhance the extraction of desirable compounds, leading to a fuller flavor profile. Conversely, water that is too soft or too hard can hinder optimal extraction, resulting in a flat or overly bitter cup. The Specialty Coffee Association (SCA) often recommends specific water parameters, including hardness, alkalinity, and pH, highlighting the scientific consensus on water’s profound impact on the final brew. While TDS provides a quantifiable measure of concentration, it offers only a partial glimpse into the complex chemical composition, as the nature of these dissolved solids varies dramatically.
The Unseen Orchestra: Identifying Coffee’s Volatile and Non-Volatile Constituents
The true marvel of coffee lies within this small percentage of dissolved solids, which house the compounds responsible for its color, taste, and aroma. Advanced analytical techniques, particularly various forms of chromatography such as Gas Chromatography-Mass Spectrometry (GC-MS) and High-Performance Liquid Chromatography (HPLC), have allowed scientists to separate and identify individual chemical components. These analyses routinely reveal hundreds, and in some cases, thousands of different molecules. However, the scientific journey is far from complete; many of these compounds remain unknown or uncharacterized, posing a significant challenge to fully understanding coffee’s complexity.
A notable aroma extract dilution analysis (AEDA) conducted in 2002 by Sanz, Czerny, Cid, and Schieberle highlighted this persistent enigma, revealing that 13 of the 40 key odorants in coffee had yet to be identified [1]. This phenomenon is often attributed to several factors: some compounds are present in extremely low concentrations, making their isolation and structural elucidation difficult; others possess large, complex molecular structures that complicate analysis. The ongoing quest to identify these unknown constituents underscores the dynamic nature of coffee research.
Early attempts to synthetically mimic coffee further illustrate its intricate nature. A 1996 study by Semmelroch and Grosch demonstrated that a blend of just 22 key volatile compounds could produce "model" coffees described as "clearly coffee-like" by assessors [2]. While a significant achievement, the phrase "clearly coffee-like" suggests a noticeable divergence from the genuine article. This finding reinforces the scientific consensus that the true, nuanced aroma and flavor of coffee depend on a far greater number of compounds, many interacting synergistically, to achieve its characteristic depth and richness. The pursuit of a perfect synthetic coffee remains an elusive goal, indicating that the natural brew offers an unparalleled sensory experience.
A Journey Through the Senses: Aroma, Taste, and Mouthfeel
The compounds within coffee can be broadly categorized into volatile and non-volatile groups, each playing distinct roles in the sensory experience. Volatile compounds, characterized by their ability to vaporize at ambient temperatures, are responsible for coffee’s complex aroma. These molecules waft into the nose through orthonasal olfaction (direct inhalation) and retronasal olfaction (from the mouth to the nasal cavity during consumption), triggering a cascade of olfactory receptors. Coffee’s aromatic profile is incredibly diverse, encompassing notes ranging from floral and fruity (esters, aldehydes), nutty and chocolatey (pyrazines, alkylfurans), to smoky and roasty (phenols, thiols). For instance, 2-furfurylthiol is a potent odorant often associated with roasted, coffee-like, and sulfurous notes, while various pyrazines contribute to the quintessential nutty and roasty character.
Non-volatile compounds, on the other hand, primarily contribute to the tastes perceived on the tongue and the physical sensation of mouthfeel. The human tongue detects five basic tastes: bitterness, sweetness, acidity, saltiness, and umami.

- Bitterness in coffee is predominantly attributed to compounds like chlorogenic acids (CGAs) and their breakdown products (e.g., quinides) formed during roasting, as well as caffeine, though caffeine’s contribution to overall bitterness is often overstated compared to CGAs.
- Acidity, a highly prized characteristic in specialty coffee, stems from various organic acids such as citric, malic, lactic, and acetic acids. The balance and type of acidity can vary significantly based on coffee origin, processing method (e.g., washed coffees often exhibit brighter acidity), and roast level.
- Sweetness is derived from simple sugars (glucose, fructose, sucrose) and soluble oligosaccharides, which are partially caramelized during roasting.
- Saltiness and umami notes, though less prominent, can be contributed by certain minerals and amino acids.
- Astringency, a drying sensation in the mouth, is typically caused by polyphenols, often referred to as tannins, which can bind with salivary proteins.
Beyond taste, non-volatile compounds and insolubles contribute significantly to coffee’s body and texture. Lipids (oils) and tiny suspended particles, particularly those present in unfiltered brews like French press, impart a viscous, rich mouthfeel, contributing to what is often described as "body." The interplay between these volatile and non-volatile components creates the holistic and deeply satisfying experience of a cup of coffee.
The Paradox of Potency: Trace Compounds and Odor Activity Values
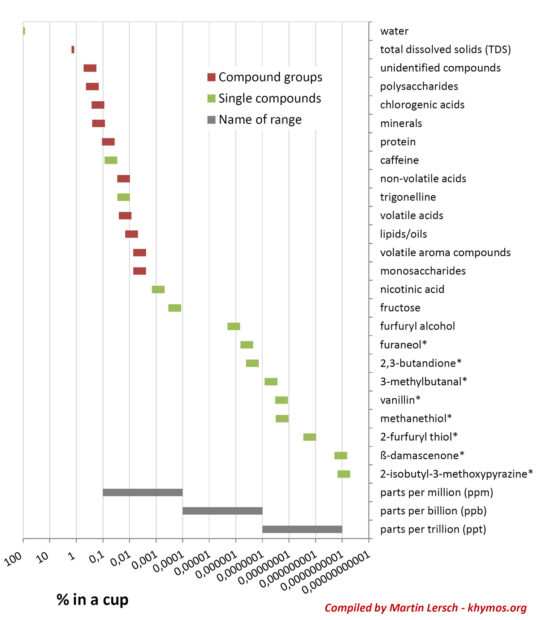
One of the most remarkable aspects of coffee chemistry is the disproportional impact of compounds present in minute quantities. While major components like water and total dissolved solids are abundant, the overall flavor profile is profoundly shaped by trace compounds existing at concentrations spanning more than nine orders of magnitude, from percentages down to parts per trillion (ppt). This logarithmic scale of concentration highlights a critical concept in flavor science: a compound’s concentration does not directly correlate with its sensory impact.
This phenomenon is explained by the concept of Odor Activity Value (OAV), which is calculated by dividing a compound’s concentration by its individual odor threshold – the minimum concentration at which it can be detected by the human nose. A high OAV indicates a significant sensory contribution even at very low concentrations, as the human nose possesses extraordinary sensitivity to certain molecules.
A prime example of such a potent trace compound is β-damascenone. Despite being present in coffee at concentrations as low as 1 part per trillion (ppt), β-damascenone is considered one of the top five key odorants in coffee [1]. Its profound impact on flavor is a testament to its extremely low odor threshold. β-damascenone belongs to a family of compounds known as rose ketones, first discovered in rose oil in the 1960s. The name "damascenone" itself is derived from the Damask rose (Rosa × damascena), where these captivating aromatics were initially identified.
The sensory profile of β-damascenone is remarkably broad and appealing. The Good Scents Company, a leading resource in flavor and fragrance chemistry, lists its odors and flavors as encompassing "fruity, flowery, sweet, blackcurrant, apple, tobacco, tea, rum, woody, honey, tropical, rose, jammy, plum, apricot." This rich bouquet of descriptors illustrates why β-damascenone is such a vital component of coffee’s complex aroma. Its significance extends beyond the culinary world; the flavor and fragrance company Firmenich pioneered its use, and β-damascenone, alongside other rose ketones, became a cornerstone in iconic perfumes, most famously in Dior’s "Poison." The recognition of its pivotal role in both perfumery and coffee flavor underscores the universal appeal and potent sensory properties of this remarkable molecule. The challenge of accurately measuring odor thresholds and quantifying compounds at such ultra-low concentrations remains a frontier in analytical chemistry, suggesting that even more key odorants in coffee may await discovery.
The Science of Solvents: Polar, Non-Polar, and Extraction Efficiency
The effectiveness of water as a solvent in coffee brewing is intrinsically linked to the polarity of the compounds being extracted. Chemical compounds can be broadly classified as polar or non-polar. Water itself is a highly polar solvent, meaning it readily dissolves other polar compounds through hydrogen bonding and electrostatic interactions – a principle often summarized as "like dissolves like."
In coffee, polar compounds include acids, minerals, proteins, and carbohydrates (sugars and soluble oligosaccharides). Due to their high solubility in water, more than 75% of these polar compounds are typically extracted during the brewing process [2]. These components contribute significantly to coffee’s acidity, sweetness, and the overall mineral balance.
Conversely, non-polar compounds, such as volatile aroma compounds (many of which are lipophilic) and lipids (oils), have very low solubility in water. As a result, standard water-based brewing methods typically extract only 10% to 30% of these non-polar constituents [2]. This differential extraction rate is crucial for understanding the final flavor profile. For instance, brewing methods that allow for greater retention of oils, such as a French press, often result in a coffee with a heavier body due to the presence of more non-polar lipids.
This inherent selectivity of water opens avenues for exploring alternative extraction methods. The use of less polar solvents, such as ethanol, could potentially unlock a different spectrum of compounds, particularly a higher proportion of non-polar aromatics and lipids that remain largely unextracted by water. While purely speculative for mainstream consumption, such experimental approaches could lead to novel coffee concentrates, essences, or even non-alcoholic coffee spirits with unique sensory profiles. However, this also presents significant challenges related to safety, regulation, cost, and consumer acceptance, alongside the potential for extracting undesirable off-flavors.
The Limitations of Metrics: Why the Nose Knows Best
While Total Dissolved Solids (TDS) measurements provide a valuable indicator of coffee strength, they are remarkably poor predictors of the actual aroma and flavor complexity in a cup of coffee. This limitation stems from several factors: the large difference in extraction yields between polar and non-polar compounds, the vastly different odor thresholds of individual molecules, and the sheer number of compounds that contribute to the overall sensory experience. A TDS reading might tell you the total amount of "stuff" in your coffee, but it cannot differentiate between the impact of a plentiful, mildly flavored sugar and a scarce, intensely aromatic β-damascenone.
The human sensory system, particularly the nose, remains an unparalleled analytical detector. Its ability to discern, interpret, and integrate the signals from hundreds of volatile compounds simultaneously allows for a holistic perception of flavor that no single chemical metric can replicate. This is why sensory evaluation, led by trained professionals in formal "cupping" sessions, remains the gold standard for assessing coffee quality and profiling. These experts can identify subtle nuances, defects, and desirable characteristics that are beyond the scope of current laboratory instruments. The intricate interplay of science and art in coffee is thus underscored: scientific analysis provides the foundational understanding, but the human palate ultimately judges the wonder of the extraction.
Broader Impact and Industry Implications
The deepening scientific understanding of coffee’s molecular composition has profound implications across the entire coffee value chain. For coffee producers and roasters, this knowledge enables more precise control over processing and roasting parameters. Roasters can tailor their profiles to enhance specific precursor compounds, unlocking desired flavor notes that are inherent to particular origins and bean varieties. This scientific insight supports the emphasis on traceability and single-origin coffees prevalent in the specialty coffee movement.
For baristas and brewers, an understanding of extraction dynamics, including the differential solubility of polar and non-polar compounds, empowers them to optimize brewing methods. Adjustments to grind size, water temperature, brew time, and pressure can selectively influence which compounds are extracted, allowing for greater customization and consistency in the final cup. This scientific grounding elevates brewing from an intuitive craft to an informed discipline.
For consumers, this wealth of information fosters a deeper appreciation for the complexity and craftsmanship behind their daily brew. It encourages informed choices and an exploration of diverse flavor profiles, moving beyond mere caffeine delivery to a pursuit of sensory pleasure. Moreover, ongoing research into coffee’s chemical makeup contributes to innovation, driving the development of new products, from enhanced instant coffees to novel coffee-based beverages that capture specific aromatic or taste elements.
The Ongoing Quest: Challenges and Future Directions
Despite significant advancements in analytical chemistry, the complete unraveling of coffee’s molecular tapestry remains an ongoing quest. Challenges persist in identifying the remaining unknown compounds, particularly those present at ultra-trace levels, and in standardizing sensory evaluation across individuals due to genetic variations in taste and smell perception. Furthermore, coffee is a dynamic system, with its chemical composition evolving during storage and brewing, adding another layer of complexity.
Future research directions are poised to push these boundaries even further. The integration of advanced computational models, including artificial intelligence and machine learning, may soon enable predictive flavor modeling, correlating chemical fingerprints with sensory outcomes. Precision extraction technologies, potentially utilizing novel solvents or advanced membrane separation techniques, could revolutionize how coffee concentrates are produced, offering unprecedented control over flavor profiles. The burgeoning field of personalized nutrition might even see coffee tailored to individual genetic predispositions for taste and smell. Ultimately, the scientific exploration of coffee’s chemical wonders promises not only to deepen our understanding of this beloved beverage but also to continually enhance the global coffee experience for generations to come.
References
[1] Sanz, C.; Czerny, M.; Cid, C.; Schieberle, P. Eur. Food. Res. Technol., 2002, 214, 299 (DOI: 10.1007/s00217-001-0459-9)
[2] Semmelroch, P.; Grosch, W. J. Agric. Food Chem. 1996, 44, 537 (DOI: 10.1021/jf9505988)
[3] Belitz, H.-D.; Grosch, W.; Schieberle, P. (ed.), 2004, Food Chemistry (3rd edition) Springer.
[4] Amanpour, A.; Selli, S. J. Food Proc. Pres. 2016, 40, 1116 (DOI: 10.1111/jfpp.12692)
[5] Batali, M. E.; Frost, S. C.; Lebrilla, C. B.; Ristenpart, W. D.; Guinard, J.-X. J. Sci. Food Agric. 2020, 7, 2953 (DOI: 10.1002/jsfa.10323)