A seemingly simple beverage, coffee, is in fact a highly complex chemical matrix, with over 98.5% of a typical black brew being water. Yet, within the remaining 1.2-1.5% of total dissolved solids (TDS)—translating to 12-15 grams of extracted compounds per liter from approximately 60 grams of coffee grounds—lies a staggering array of hundreds, if not thousands, of distinct molecules. This intricate chemical profile is precisely what confers coffee its signature color, diverse tastes, and captivating aromas, a testament to nature’s complexity and the transformative power of roasting and brewing.
The scientific journey into understanding coffee’s composition has been a persistent quest, driven by both academic curiosity and the industry’s pursuit of perfection. Researchers employing sophisticated chromatographic techniques, such as gas chromatography-mass spectrometry (GC-MS) and aroma extract dilution analysis (AEDA), have painstakingly separated and identified many of these individual chemical components. However, the sheer number and the incredibly low concentrations of some key flavor compounds present significant challenges. A notable AEDA study from 2002 revealed that out of 40 key odorants identified in coffee, a remarkable 13 remained unknown to science at the time. This highlights the ongoing frontier of coffee chemistry, where molecules, often present in concentrations as low as parts per trillion, exert profound influence on the overall sensory experience.
The Elusive Essence: A Deep Dive into Coffee’s Composition
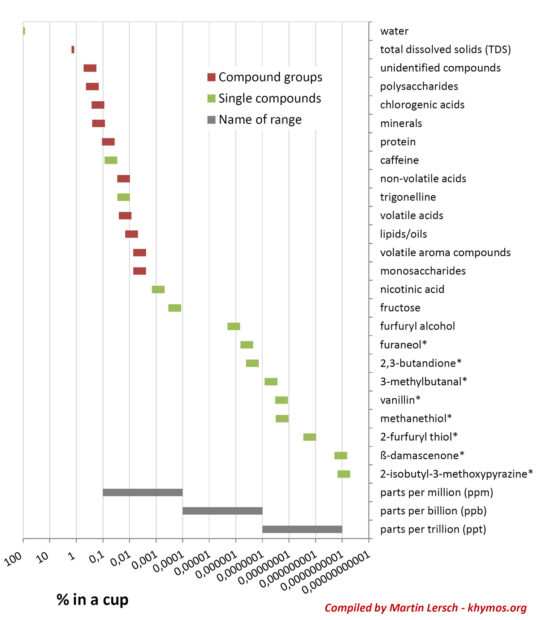
The myriad compounds found in a cup of coffee can broadly be categorized into volatile and non-volatile components. Volatile compounds, characterized by their ability to readily evaporate and reach the olfactory receptors in the nose, are primarily responsible for coffee’s diverse aromas. These include a vast spectrum of chemical classes such as pyrazines (contributing nutty, roasted notes), furans (caramel, sweet), thiols (savory, roasted), and aldehydes (fruity, green). In contrast, non-volatile compounds contribute to the tastes perceived on the tongue—bitterness, sweetness, acidity, and astringency—as well as the body and texture of the beverage. This category encompasses carbohydrates (various sugars and soluble oligosaccharides), organic acids (e.g., chlorogenic acids, quinic acid, acetic acid), minerals, proteins, lipids, and the well-known stimulant, caffeine. Beyond these identified groups, a substantial portion remains "unidentified," likely contributing significantly to coffee’s color and bitterness.
The intricate dance of these components begins long before brewing. The roasting process is a crucial stage where chemical precursors within the green coffee bean undergo complex transformations, notably through the Maillard reaction and Strecker degradation. These reactions generate hundreds of new volatile compounds, shaping the distinct aroma profile of a roasted bean. For instance, chlorogenic acids, abundant in green coffee, largely degrade during roasting to form quinic and caffeic acids, contributing to bitterness and acidity. Sugars caramelize, producing sweet and toasted notes, while lipids, though largely non-soluble, encapsulate and release volatile aroma compounds during brewing, contributing to the coffee’s mouthfeel and flavor persistence.
A Scientific Chronology: Deciphering Coffee’s Code
The scientific understanding of coffee has evolved significantly over centuries. Early investigations primarily focused on identifying its most prominent components. Caffeine, for example, was isolated and identified as early as 1819 by German chemist Friedlieb Ferdinand Runge, marking one of the first major breakthroughs. However, the true complexity of coffee’s flavor and aroma remained largely mysterious until the mid-20th century.
The advent of sophisticated analytical techniques, particularly gas chromatography coupled with mass spectrometry (GC-MS) in the 1960s, revolutionized flavor chemistry. These tools allowed scientists to separate and identify minute quantities of volatile compounds, opening the door to a deeper understanding of coffee’s aroma profile. This era marked a shift from simply identifying major components to meticulously cataloging the subtle nuances that define sensory experience.

In 1996, a landmark study by Semmelroch and Grosch demonstrated the immense challenge of replicating coffee flavor synthetically. By combining 22 key volatile compounds, they created "model" coffees that assessors described as "clearly coffee-like." While a significant achievement, this observation underscored that 22 compounds were far from sufficient to perfectly mimic the richness and depth of natural Arabica and Robusta coffees. It highlighted that the true essence of coffee flavor relies on a much broader and more intricate symphony of molecules.
Further deepening this understanding, the 2002 AEDA study by Sanz, Czerny, Cid, and Schieberle pinpointed 40 "key odorants" in coffee, reinforcing the notion that flavor is not simply a sum of its parts but rather an emergent property of complex interactions. The discovery that 13 of these crucial odorants were previously unknown served as a powerful reminder of the vast uncharted territory within coffee chemistry, spurring further research into low-concentration, high-impact compounds.
The Power of Perception: Odor Activity Values and Beta-Damascenone
One of the most profound insights in flavor science is the concept of Odor Activity Value (OAV). OAV is calculated by dividing the concentration of a compound by its specific odor threshold—the minimum concentration required for a compound to be perceived by the human nose. This metric elegantly explains why a compound present at an infinitesimal concentration, say 0.0000000001%, can have as significant an impact on overall flavor as another compound present at 0.01%. The sensitivity of our olfactory system varies dramatically for different molecules.
A prime example of such a potent, low-concentration odorant in coffee is β-damascenone. This remarkable compound, present in concentrations down to 1 part per trillion (ppt), is considered one of the top five most impactful odorants in coffee. Its contribution is often described with notes of rose, blackcurrant, plum, and honey—a complex profile that adds significant depth and fruitiness to coffee’s aroma. β-damascenone belongs to a group known as rose ketones, first discovered in rose oil in the 1960s. Its name is derived from the Damask rose (Rosa × damascena), a flower renowned for its intoxicating fragrance. The flavor and fragrance industry quickly recognized its potential; Firmenich, a leading company, famously incorporated β-damascenone and other rose ketones into Dior’s iconic perfume "Poison," demonstrating its versatility and powerful aromatic properties across different sensory applications.
Beyond β-damascenone, other low-concentration compounds play critical roles. Sulfur-containing compounds, for instance, contribute savory, roasted, and sometimes meaty notes, even at ppb levels. Pyrazines provide distinctive roasted, nutty, and bready aromas, while certain aldehydes contribute fruity or green nuances. The intricate interplay of these high-OAV compounds, even when present in minute quantities, orchestrates the rich and multi-faceted aroma that coffee enthusiasts cherish.
Extraction Dynamics: Polar, Non-Polar, and the Brewing Challenge
The act of brewing coffee is fundamentally an extraction process, where hot water acts as a solvent to draw out compounds from ground coffee beans. The efficiency and selectivity of this extraction are heavily influenced by the polarity of the compounds. Polar compounds, such as acids, minerals, proteins, and carbohydrates, are highly soluble in water. Consequently, typical brewing methods can extract more than 75% of these water-loving molecules. These components significantly contribute to coffee’s acidity, sweetness, and overall body.
In contrast, non-polar compounds, including many volatile aroma compounds and lipids, exhibit low solubility in water. As a result, only about 10-30% of these compounds are typically extracted during conventional water-based brewing. This differential extraction poses a significant challenge and opportunity for coffee professionals. For instance, the lipids, while poorly water-soluble, form an emulsion that contributes to the crema in espresso and the rich mouthfeel of certain brews. Research into alternative extraction methods, such as using less polar solvents like ethanol, is exploring ways to unlock different facets of coffee’s flavor profile, potentially yielding novel sensory experiences. Such experimental approaches, while not mainstream for consumption, offer valuable insights into the full chemical potential of the coffee bean.
Beyond TDS: The Limitations of Simple Metrics
While Total Dissolved Solids (TDS) measurements provide a convenient and widely used metric for coffee strength, they offer an incomplete picture of a brew’s quality and flavor complexity. A TDS reading, typically ranging from 1.2% to 1.5% for black coffee, primarily reflects the concentration of the most abundant soluble compounds—chiefly carbohydrates, acids, and caffeine. It fails to account for the disproportionate impact of low-concentration, high-OAV aroma compounds.
The problem lies in the lack of a direct correlation between a compound’s concentration and its sensory impact, as well as the varying extraction rates of different compound classes. A high TDS might indicate a strong brew, but it provides little information about the balance of sweetness, acidity, bitterness, or the nuanced aromatic notes that define a truly exceptional cup. For example, over-extraction can lead to high TDS but also an influx of undesirable bitter or astringent compounds. Conversely, an under-extracted coffee might have a lower TDS but lack the desired complexity.
Therefore, while TDS remains a valuable tool for consistency in brewing, it is a poor predictor of the actual aroma and flavor perceived by a discerning palate. The human nose, with its unparalleled ability to detect and interpret complex mixtures of volatile compounds, remains the most sophisticated and irreplaceable "analytical detector" in the world of coffee. This emphasizes why sensory evaluation, led by expert tasters and baristas, continues to be paramount in assessing coffee quality and guiding improvements in roasting and brewing.
Implications for Industry and Innovation
The deepening understanding of coffee chemistry has profound implications across the entire coffee value chain. For the specialty coffee industry, this scientific insight empowers roasters and baristas to manipulate profiles with unprecedented precision. Knowledge of specific compounds and their precursors allows for tailored roasting strategies to highlight desirable notes (e.g., fruitiness, floral, chocolate) and mitigate less favorable ones. Baristas can fine-tune extraction parameters—grind size, water temperature, brew time, and even water mineral composition—to selectively extract compounds and achieve specific flavor outcomes, pushing the boundaries of what a cup of coffee can be.
In health and nutrition, research into coffee’s chemical makeup continues to uncover its multifaceted impact on the human body. Beyond caffeine, compounds like chlorogenic acids, melanoidins, and diterpenes (cafestol, kahweol) are studied for their antioxidant, anti-inflammatory, and other potential health benefits. This has led to discussions about brewing methods (e.g., filtered vs. unfiltered coffee) affecting the concentration of certain compounds and their subsequent physiological effects.
The future of coffee innovation also stands to benefit. This scientific exploration could lead to the development of more advanced brewing equipment capable of highly precise and selective extractions. It could inform the creation of "designer" coffee blends, formulated not just on bean origin but on a desired molecular profile. While a truly "synthetic" coffee that perfectly replicates the natural complexity remains a distant prospect, the ongoing research into individual key odorants could enhance flavor profiles in coffee products or even inform the development of novel food and beverage applications. Furthermore, understanding the chemical transformations throughout the coffee process can contribute to more sustainable practices, from optimized processing techniques to reducing waste by extracting valuable compounds from spent grounds.
The Unending Quest for the Perfect Cup
The journey into the molecular world of coffee is far from over. Each new analytical technique, each newly identified compound, adds another layer to our appreciation of this ubiquitous beverage. The balance between scientific rigor and the nuanced art of sensory perception remains central to the coffee experience. As researchers continue to unravel the secrets held within coffee’s minute chemical constituents, enthusiasts and professionals alike will continue to explore the "wonders of extraction," driven by the unending quest for the perfect cup. The little red fox in the coffee cup, perhaps, symbolizes the elusive, wild, and incredibly complex nature of the brew, challenging us to look beyond the obvious and delve into its microscopic marvels.