The daily ritual of coffee consumption, often taken for granted, conceals an astonishing world of chemical complexity, a symphony of hundreds, if not thousands, of distinct molecules contributing to its unparalleled aroma, taste, and body. While enthusiasts meticulously refine parameters like brew ratio, grind size, and water temperature, the true magic unfolds at the molecular level, where a mere fraction of the beverage’s composition dictates its profound sensory impact. This exploration delves into the intricate chemistry of a brewed cup, revealing why this seemingly simple drink is, in fact, one of the most chemically sophisticated beverages on Earth.
The Invisible Orchestra: Hundreds of Molecules in Every Sip
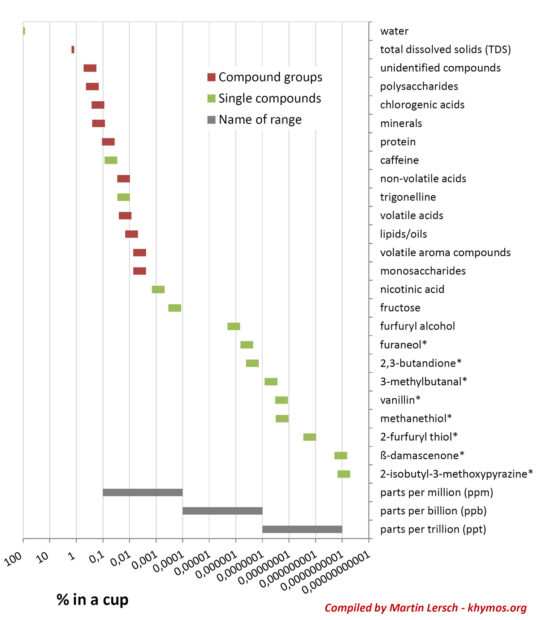
At its core, black coffee is overwhelmingly water, constituting more than 98.5% of the final beverage. The remaining 1.2% to 1.5% comprises total dissolved solids (TDS), meaning that approximately 12 to 15 grams of various compounds are extracted from roughly 60 grams of coffee grounds to produce a single liter of brewed coffee. This seemingly small percentage is a rich tapestry of chemical compounds responsible for coffee’s characteristic color, flavor, and fragrance. Advanced analytical techniques, particularly various forms of chromatography such as Gas Chromatography-Mass Spectrometry (GC-MS) and Liquid Chromatography-Mass Spectrometry (LC-MS), have allowed scientists to peel back the layers of this complexity. These methods separate individual chemical components, revealing a vast molecular landscape. Research suggests that coffee contains hundreds, and potentially thousands, of different molecules. While many have been identified and characterized, a significant number remain elusive, presenting an ongoing challenge to flavor scientists. For instance, an aroma extract dilution analysis (AEDA) conducted in 2002 notably reported that 13 of the 40 most crucial odorants contributing to coffee’s distinctive aroma were still unknown.
The difficulty in identifying these compounds often stems from their extremely low concentrations, making it challenging to isolate sufficient quantities for full structural elucidation. Large molecular structures also complicate the analytical process. This molecular complexity explains why efforts to artificially replicate coffee flavor have met with limited success. A 1996 study, for example, demonstrated that a "model" coffee concocted from 22 key volatile compounds could be described as "clearly coffee-like" by sensory assessors, mimicking Arabica and Robusta varieties. However, the nuance and depth of a natural brew remained out of reach, underscoring that 22 compounds are a mere prelude to coffee’s full aromatic orchestra. This ongoing scientific quest highlights the profound artistry of nature in crafting a beverage that continues to defy complete synthetic imitation.
Unveiling the Unknown: The Quest for Coffee’s Signature Aromas
The myriad compounds in coffee can be broadly categorized into volatile and non-volatile components. Volatile compounds, characterized by their low boiling points, readily evaporate and travel to the olfactory receptors in the nose, contributing the rich and diverse aroma profile. Conversely, non-volatile compounds interact with taste receptors on the tongue, influencing the primary tastes of bitterness, sweetness, acidity, and astringency. Some molecules can influence both aroma and taste, though typically, the taste threshold for a compound is considerably higher than its odor threshold. Beyond these fundamental tastes, non-volatile substances like oils and microscopic insoluble particles contribute significantly to coffee’s body and texture, impacting mouthfeel.
A deeper dive into the soluble components reveals several major groups:

- Carbohydrates: Sugars and soluble oligosaccharides contribute to sweetness and body.
- Acids: A complex array of organic acids (e.g., chlorogenic acids, citric acid, malic acid, quinic acid, acetic acid) dictates much of coffee’s characteristic acidity and brightness, which varies significantly with bean origin and roast level.
- Minerals/Salts: Trace elements like potassium, magnesium, and calcium are extracted from the bean and water, influencing overall taste perception and contributing to the buffering capacity of the brew.
- Proteins: While many proteins are denatured during roasting, some soluble peptides and amino acids remain, contributing to body and potentially taste.
- Lipids: Coffee oils, particularly abundant in espresso, contribute to mouthfeel, crema, and can carry volatile aroma compounds.
- Caffeine: The most famous compound, caffeine, is a major contributor to coffee’s bitterness and its stimulant effects.
- Volatile Aroma Compounds: This is the most diverse group, responsible for the vast array of descriptors used by coffee connoisseurs, from floral and fruity to nutty, chocolatey, earthy, and roasted.
- Unidentified Compounds: A substantial portion of extracted solids still comprises compounds whose exact chemical structures and sensory contributions are yet to be fully elucidated. These often contribute to color and general bitterness.
The Power of Potency: When Less is More in Flavor
The sheer range of concentrations of these compounds within a cup of coffee is staggering, spanning more than nine orders of magnitude, from percentages down to parts per trillion. This logarithmic scale underscores a critical principle in flavor science: concentration alone does not dictate impact. Our olfactory system possesses an extraordinary sensitivity to certain molecules, meaning a compound present in minute quantities can have a disproportionately significant effect on the overall flavor profile. This concept is quantified by the Odor Activity Value (OAV), calculated as the ratio of a compound’s concentration to its odor threshold. A high OAV indicates a strong sensory contribution, even at very low concentrations.
A prime example of such a potent odorant is β-damascenone. Present in coffee at concentrations as low as 1 part per trillion (ppt), β-damascenone is nevertheless considered one of the top five most impactful odorants in coffee, according to a prominent study. Its sensory descriptors are complex and highly desirable, encompassing notes of fruity (blackcurrant, apple), floral (rose), honey, and tobacco. This molecule belongs to a class of compounds known as rose ketones, first identified in rose oil in the 1960s. The name "damascenone" itself derives from the Damask rose (Rosa × damascena), a flower famed for its exquisite fragrance. The flavor and fragrance industry quickly recognized the potential of rose ketones; Firmenich, a leading company in the field, pioneered their application. Notably, β-damascenone, in concert with other rose ketones, became a pivotal component in Dior’s iconic perfume, Poison, highlighting its versatility and profound aromatic power across different sensory domains. The ability of the human nose to detect and integrate such minute concentrations into a holistic flavor experience remains one of biology’s most remarkable feats.
The Dance of Solvents: Polar, Non-Polar, and Extraction Dynamics
The process of brewing coffee is essentially an act of selective extraction, where water acts as a solvent to draw out compounds from the ground coffee. The efficiency of this extraction is heavily dependent on the polarity of both the solvent (water) and the compounds within the coffee grounds. Compounds can be broadly classified as polar or non-polar.
- Polar Compounds: This group includes acids, minerals, proteins, and carbohydrates. These compounds share a common characteristic: they are highly soluble in water, which is a polar solvent. Consequently, when brewing coffee with water, more than 75% of these polar compounds are typically extracted. They contribute significantly to coffee’s acidity, sweetness, and overall body.
- Non-Polar Compounds: This category encompasses volatile aroma compounds and lipids (oils). These compounds have low solubility in water. As a result, water typically extracts only 10-30% of the non-polar compounds present in coffee grounds. This differential extraction efficiency is crucial in understanding coffee flavor. While water is excellent at dissolving the bulk of the solids, it is less efficient at capturing the full spectrum of non-polar aroma compounds that are essential to the coffee’s fragrance. This insight opens avenues for experimental brewing, where less polar solvents like ethanol might be considered to explore alternative dimensions of coffee extraction, though such approaches are currently confined to research rather than mainstream consumption.
Beyond the Numbers: Why TDS Falls Short
Given the immense chemical diversity and the complex interplay of concentrations and sensory thresholds, it becomes clear why Total Dissolved Solids (TDS) measurements, while a useful indicator of coffee strength, are fundamentally limited in predicting the actual aroma and flavor profile of a cup. TDS measures the total mass of dissolved solids, but it provides no information about the identity or sensory contribution of these solids.
The primary reason for TDS’s inadequacy as a flavor predictor lies in the disparate extraction rates of various compounds and their vastly different odor thresholds. Compounds that significantly influence TDS (e.g., carbohydrates, acids, caffeine) are often extracted at different rates than the trace volatile compounds responsible for much of the aroma. Moreover, a compound present in high concentration might contribute little to flavor if its odor threshold is high, while a compound at minuscule concentrations can be a flavor powerhouse if its odor threshold is exceptionally low (like β-damascenone). Therefore, two coffee brews with identical TDS readings could possess drastically different flavor profiles due to variations in the specific blend of extracted compounds.
While TDS remains the simplest and most accessible method for gauging coffee strength and ensuring consistency in brewing parameters, relying solely on it to assess flavor quality is akin to judging a symphony by the total weight of the instruments. For most enthusiasts and professionals, the human nose, a superb analytical detector honed over millennia, remains the ultimate arbiter of coffee’s nuanced wonders, guiding the ongoing exploration of its extraction complexities.
The Broader Implications: From Roastery to Research Lab
The profound understanding of coffee’s chemical makeup has far-reaching implications across the entire coffee value chain. For roasters, this knowledge is instrumental in fine-tuning roasting profiles. Different roast levels dramatically alter the chemical composition, developing new aromatic compounds through reactions like the Maillard reaction and Strecker degradation, while degrading others. Understanding which precursors are present and how they react allows for precise control over flavor development. For baristas and brewers, it emphasizes that optimal extraction goes beyond merely achieving a certain TDS. It involves carefully balancing the extraction of polar and non-polar compounds to achieve a harmonious and desirable flavor balance.
In the research and development sector, the ongoing discovery of new compounds and the elucidation of their sensory contributions continue to push the boundaries of coffee science. This leads to innovations in coffee processing, breeding new varieties with enhanced flavor precursors, and developing more sophisticated quality control mechanisms that move beyond simple physical parameters. For consumers, this scientific insight enriches the appreciation of coffee, transforming it from a mere morning stimulant into a complex gastronomic experience. Understanding the "why" behind different flavor notes allows for a more informed and engaging exploration of coffee origins, varietals, and brewing methods. The continuous quest to unravel coffee’s molecular secrets ensures that the world’s most beloved beverage will remain a fascinating subject of both scientific inquiry and sensory delight for generations to come.
Looking Ahead: The Ever-Evolving Science of Coffee
The journey to fully understand the chemical marvel that is coffee is far from over. Each new analytical technique, each newly identified compound, adds another piece to the intricate puzzle. As technology advances, allowing for the detection and quantification of compounds at even lower concentrations, it is highly probable that additional key odorants and flavor compounds will be discovered, further enriching our understanding of this complex beverage. The interplay between genetics, terroir, processing, roasting, and brewing parameters creates an almost infinite array of chemical possibilities, each contributing to the unique character of a cup. Ultimately, the science of coffee reinforces the idea that even in the most familiar of daily rituals, there lies an extraordinary depth of complexity, inviting continuous exploration and appreciation.









